KDF1, a Novel Tumor Suppressor in Clear Cell Renal Cell Carcinoma
- PMID: 34136411
- PMCID: PMC8201614
- DOI: 10.3389/fonc.2021.686678
KDF1, a Novel Tumor Suppressor in Clear Cell Renal Cell Carcinoma
Abstract
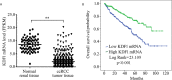
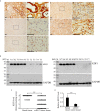
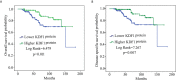
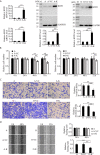
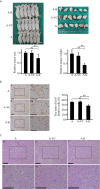
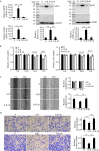
KDF1 has been identified as a key regulator of epidermal proliferation and differentiation, but it is unknown whether KDF1 is involved in the pathogenesis of malignancy. No study has reported the expression and function of KDF1 in renal cancer. To explore the pathologic significance of KDF1 in clear cell renal cell carcinoma (ccRCC), the expression level of KDF1 protein in the tumor tissue of ccRCC patients was examined by immunohistochemistry and Western blot while the expression level of KDF1 mRNA was analyzed by using the data from TCGA database. In vitro cell experiments and allogeneic tumor transplantation tests were performed to determine the effects of altered KDF1 expression on the phenotype of ccRCC cells. Both the KDF1 mRNA and protein were found to be decreasingly expressed in the tumor tissue of ccRCC patients when compared with the adjacent non-tumor control tissue. The expression level of KDF1 in the tumor tissue was found to correlate negatively with the tumor grade. Patients with higher KDF1 in the tumor tissue were found to have longer overall survival and disease-specific survival time. KDF1 was shown to be an independent factor influencing the disease-specific survival of the ccRCC patients. Overexpression of KDF1 was found to inhibit the proliferation, migration and invasion of ccRCC cells, which could be reversed by decreasing the expression of KDF1 again. ccRCC cells with KDF1 overexpression were found to produce smaller transgrafted tumors. These results support the idea that KDF1 is involved in ccRCC and may function as a tumor suppressor.
Keywords: KDF1; clear cell renal cell carcinoma; prognosis; tumor grade; tumor suppressor.
Copyright © 2021 Zheng, Gan, Yu, Ye, Yu, Xia, Zhou, Bao and Guo.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Systematic Construction and Validation of a Novel Macrophage Differentiation-Associated Prognostic Model for Clear Cell Renal Cell Carcinoma.Front Genet. 2022 Jun 14;13:877656. doi: 10.3389/fgene.2022.877656. eCollection 2022. Front Genet. 2022. PMID: 35774505 Free PMC article.
-
KDF1 Promoted Proliferation and Metastasis of Epithelial Ovarian Cancer via Wnt/Beta-Catenin Pathway: TCGA-Based Data Mining and Experimental Validation.Front Genet. 2022 Feb 25;13:808100. doi: 10.3389/fgene.2022.808100. eCollection 2022. Front Genet. 2022. PMID: 35281796 Free PMC article.
-
KDF1 Promoted Proliferation, Migration and Invasion of Lung Adenocarcinoma Cells through Activating STAT3 and AKT Pathway.Biomedicines. 2023 Dec 1;11(12):3194. doi: 10.3390/biomedicines11123194. Biomedicines. 2023. PMID: 38137415 Free PMC article.
-
Reduced expression of growth and differentiation factor-9 (GDF9) is associated with aggressive behaviour of human clear-cell renal cell carcinoma and poor patient survival.Anticancer Res. 2014 Nov;34(11):6515-20. Anticancer Res. 2014. PMID: 25368253
-
VHL mutation-mediated SALL4 overexpression promotes tumorigenesis and vascularization of clear cell renal cell carcinoma via Akt/GSK-3β signaling.J Exp Clin Cancer Res. 2020 Jun 8;39(1):104. doi: 10.1186/s13046-020-01609-8. J Exp Clin Cancer Res. 2020. PMID: 32513235 Free PMC article.
Cited by
-
Systematic Construction and Validation of a Novel Macrophage Differentiation-Associated Prognostic Model for Clear Cell Renal Cell Carcinoma.Front Genet. 2022 Jun 14;13:877656. doi: 10.3389/fgene.2022.877656. eCollection 2022. Front Genet. 2022. PMID: 35774505 Free PMC article.
-
Predictive biomarkers for metachronous gastric cancer development after endoscopic resection of early gastric cancer.Cancer Med. 2024 Aug;13(16):e70104. doi: 10.1002/cam4.70104. Cancer Med. 2024. PMID: 39171503 Free PMC article.
-
From Fruit Waste to Medical Insight: The Comprehensive Role of Watermelon Rind Extract on Renal Adenocarcinoma Cellular and Transcriptomic Dynamics.Int J Mol Sci. 2023 Oct 26;24(21):15615. doi: 10.3390/ijms242115615. Int J Mol Sci. 2023. PMID: 37958599 Free PMC article.
-
The NOTCH-RIPK4-IRF6-ELOVL4 Axis Suppresses Squamous Cell Carcinoma.Cancers (Basel). 2023 Jan 25;15(3):737. doi: 10.3390/cancers15030737. Cancers (Basel). 2023. PMID: 36765696 Free PMC article.
-
APPL1 Is a Prognostic Biomarker and Correlated with Treg Cell Infiltration via Oxygen-Consuming Metabolism in Renal Clear Cell Carcinoma.Oxid Med Cell Longev. 2023 Feb 17;2023:5885203. doi: 10.1155/2023/5885203. eCollection 2023. Oxid Med Cell Longev. 2023. PMID: 36846720 Free PMC article.
References
LinkOut - more resources
Full Text Sources

