A case of thrombocytopenia and multiple thromboses after vaccination with ChAdOx1 nCoV-19 against SARS-CoV-2
- PMID: 34137813
- PMCID: PMC8219289
- DOI: 10.1182/bloodadvances.2021004904
A case of thrombocytopenia and multiple thromboses after vaccination with ChAdOx1 nCoV-19 against SARS-CoV-2
Abstract
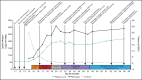
Recently, reports of severe thromboses, thrombocytopenia, and hemorrhage in persons vaccinated with the chimpanzee adenovirus-vectored vaccine (ChAdOx1 nCoV-19, AZD1222, Vaxzevria; Oxford/AstraZeneca) against severe acute respiratory syndrome coronavirus 2 have emerged. We describe an otherwise healthy 30-year-old woman who developed thrombocytopenia, ecchymosis, portal vein thrombosis, and cerebral venous sinus thrombosis the second week after she received the ChAdOx1 nCoV-19 vaccine. Extensive diagnostic workup for thrombosis predispositions showed heterozygosity for the prothrombin mutation, but no evidence of myeloproliferative neoplasia or infectious or autoimmune diseases. Her only temporary risk factor was long-term use of oral contraceptive pills (OCPs). Although both the prothrombin mutation and use of OCPs predispose to portal and cerebral vein thrombosis, the occurrence of multiple thromboses within a short time and the associated pattern of thrombocytopenia and consumption coagulopathy are highly unusual. A maximum 4T heparin-induced thrombocytopenia (HIT) score and a positive immunoassay for anti-platelet factor 4/heparin antibodies identified autoimmune HIT as a potential pathogenic mechanism. Although causality has not been established, our case emphasizes the importance of clinical awareness. Further studies of this potentially new clinical entity have suggested that it should be regarded as a vaccine-induced immune thrombotic thrombocytopenia.
© 2021 by The American Society of Hematology.
Conflict of interest statement
Conflict-of-interest disclosure: A.L.T.S. is a former employee of Novo Nordisk Biopharm and has received honoraria for lecture from Takeda and advisory board honoraria from SOBI (both not related to the present case). Z.B.H. received an unrestricted research grant from the Independent Research Fund Denmark (not related to the present case), and received travel expenses for attending a medical conference from Pfizer. C.R. has received travel expenses for attending medical conferences from Pfizer and MSD. L.K. has received an honorarium for a lecture on pneumococcal disease from MSD. J.H. receives consulting fees for Novo Nordisk on drug delivery. E.B.L. has received honoraria from Amgen for a lecture on immune thrombocytopenia (not related to present case). A.-M.H. has received an unrestricted research grant from CSL Behring (not related to the present case) and has received honoraria for lectures from CSL Behring, Bayer, Astellas, and Boehringer-Ingelheim. P.K. has received honoraria for lectures from uniQure and Takeda and travel expenses for attendance at medical conferences from uniQure. The remaining authors declare no competing financial interests.
Figures
References
-
- European Medicines Agency. COVID-19 Vaccine AstraZeneca: benefits still outweigh the risks despite possible link to rare blood clots with low blood platelets. Available at: https://www.ema.europa.eu/en/news/covid-19-vaccine-astrazeneca-benefits-...
-
- Lo GK, Juhl D, Warkentin TE, Sigouin CS, Eichler P, Greinacher A.. Evaluation of pretest clinical score (4 T’s) for the diagnosis of heparin-induced thrombocytopenia in two clinical settings. J Thromb Haemost. 2006;4(4):759-765. - PubMed
-
- Greinacher A, Selleng K, Warkentin TE.. Autoimmune heparin-induced thrombocytopenia. J Thromb Haemost. 2017;15(11):2099-2114. - PubMed
-
- Juhl D, Eichler P, Lubenow N, Strobel U, Wessel A, Greinacher A.. Incidence and clinical significance of anti-PF4/heparin antibodies of the IgG, IgM, and IgA class in 755 consecutive patient samples referred for diagnostic testing for heparin-induced thrombocytopenia. Eur J Haematol. 2006;76(5):420-426. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

