Illustrating potential effects of alternate control populations on real-world evidence-based statistical analyses
- PMID: 34142018
- PMCID: PMC8206406
- DOI: 10.1093/jamiaopen/ooab045
Illustrating potential effects of alternate control populations on real-world evidence-based statistical analyses
Abstract
Objective: Case-control study designs are commonly used in retrospective analyses of real-world evidence (RWE). Due to the increasingly wide availability of RWE, it can be difficult to determine whether findings are robust or the result of testing multiple hypotheses.
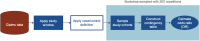
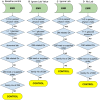
Materials and methods: We investigate the potential effects of modifying cohort definitions in a case-control association study between depression and type 2 diabetes mellitus. We used a large (>75 million individuals) de-identified administrative claims database to observe the effects of minor changes to the requirements of glucose and hemoglobin A1c tests in the control group.
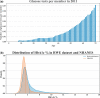
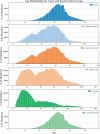
Results: We found that small permutations to the criteria used to define the control population result in significant shifts in both the demographic structure of the identified cohort as well as the odds ratio of association. These differences remain present when testing against age- and sex-matched controls.
Discussion: Analyses of RWE need to be carefully designed to avoid issues of multiple testing. Minor changes to control cohorts can lead to significantly different results and have the potential to alter even prospective studies through selection bias.
Conclusion: We believe this work offers strong support for the need for robust guidelines, best practices, and regulations around the use of observational RWE for clinical or regulatory decision-making.
Keywords: association testing; informatics; phenotyping; real-world evidence.
© The Author(s) 2021. Published by Oxford University Press on behalf of the American Medical Informatics Association.
Conflict of interest statement
None declared.
Figures
Similar articles
-
The future of Cochrane Neonatal.Early Hum Dev. 2020 Nov;150:105191. doi: 10.1016/j.earlhumdev.2020.105191. Epub 2020 Sep 12. Early Hum Dev. 2020. PMID: 33036834
-
Meta-analyses using real-world data to generate clinical and epidemiological evidence: a systematic literature review of existing recommendations.Curr Med Res Opin. 2018 Dec;34(12):2125-2130. doi: 10.1080/03007995.2018.1524751. Epub 2018 Sep 25. Curr Med Res Opin. 2018. PMID: 30217138
-
Trial designs using real-world data: The changing landscape of the regulatory approval process.Pharmacoepidemiol Drug Saf. 2020 Oct;29(10):1201-1212. doi: 10.1002/pds.4932. Epub 2019 Dec 10. Pharmacoepidemiol Drug Saf. 2020. PMID: 31823482 Free PMC article. Review.
-
Improving Transparency to Build Trust in Real-World Secondary Data Studies for Hypothesis Testing-Why, What, and How: Recommendations and a Road Map from the Real-World Evidence Transparency Initiative.Value Health. 2020 Sep;23(9):1128-1136. doi: 10.1016/j.jval.2020.04.002. Value Health. 2020. PMID: 32940229
-
RWE Framework: An Interactive Visual Tool to Support a Real-World Evidence Study Design.Drugs Real World Outcomes. 2019 Dec;6(4):193-203. doi: 10.1007/s40801-019-00167-6. Drugs Real World Outcomes. 2019. PMID: 31741199 Free PMC article.
Cited by
-
Opportunities and challenges for biomarker discovery using electronic health record data.Trends Mol Med. 2023 Sep;29(9):765-776. doi: 10.1016/j.molmed.2023.06.006. Epub 2023 Jul 18. Trends Mol Med. 2023. PMID: 37474378 Free PMC article. Review.
-
Disease progression strikingly differs in research and real-world Parkinson's populations.NPJ Parkinsons Dis. 2024 Mar 13;10(1):58. doi: 10.1038/s41531-024-00667-5. NPJ Parkinsons Dis. 2024. PMID: 38480700 Free PMC article.
References
-
- Jarow JP, LaVange L, Woodcock J.. Multidimensional evidence generation and FDA regulatory decision making: defining and using “real-world” data. JAMA 2017; 318: 703–4. - PubMed
-
- Corrigan-Curay J, Sacks L, Woodcock J.. Real-world evidence and real-world data for evaluating drug safety and effectiveness. JAMA 2018; 320: 867. - PubMed
-
- The FDA and Flatiron Health Expand Real-World Data Cancer Research Collaboration | Flatiron Health. Flatiron Health. 2019. https://flatiron.com/press/press-release/the-fda-and-flatiron-health-exp... Accessed March 4, 2019.
-
- FDA. Number of Breakthrough Therapy Designation Requests Received. FDA Data Access. 2019. https://www.accessdata.fda.gov/scripts/fdatrack/view/track.cfm?program=c... Accessed September 10, 2019.
-
- FDA. CDER Drug and Biologic Accelerated Approvals Based on a Surrogate Endpoint. fda.gov. 2019. https://www.fda.gov/media/88907/download Accessed August 3, 2019.
Grants and funding
LinkOut - more resources
Full Text Sources
