Conditioned medium of IGF1-induced synovial membrane mesenchymal stem cells increases chondrogenic and chondroprotective markers in chondrocyte inflammation
- PMID: 34143208
- PMCID: PMC8255536
- DOI: 10.1042/BSR20202038
Conditioned medium of IGF1-induced synovial membrane mesenchymal stem cells increases chondrogenic and chondroprotective markers in chondrocyte inflammation
Abstract
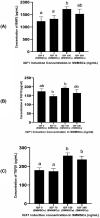
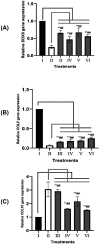
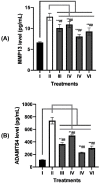
Recently, mesenchymal stem cells (MSCs) have been the most explored cells for cell therapy for osteoarthritis (OA) that can be obtained from various sources. Synovial membrane MSCs (SMMSCs) provide best potential for OA therapy, however they are not widely explored. Conditioned medium of SMMSCs (SMMSCs-CM) rich in growth factors and cytokines can inhibit apoptosis and increase chondrocytes cell proliferation. The aim of the present study was to determine growth factors content in SMMSCs-CM as well as the chondrogenic and chondroprotective markers expression in OA model after insulin-like growth factor (IGF)1-induced and non-induced SMMSCs-CM treatments. Chondrocyte cell line (CHON002) was induced by IL1β as OA model (CHON002 with IL1β (IL1β-CHON002)) and treated with SMMSCs-CM with or without IGF1 induction to determine its effectiveness in repairing OA cells model. ELISA was used to assay BMP2, fibroblast growth factor 18 (FGF18) and transforming growth factor (TGF) β1 (TGFβ1) levels in SMMSCs-CM, matrix metalloproteinase (MMP) 13 (MMP13) and a disintegrin and metalloproteinase with thrombospondin motif 4 (ADAMTS4) levels in OA cells model treated with SMMSCs-CM. RT-qPCR analyses were used to investigate the gene expression of SOX9, COL2, and COL10. CM from SMMSCs cultured and induced by IGF1 150 ng/mL was the most effective concentration for increasing the content of growth factor markers of SMMSCs-CM, which had successfully increased negative cartilage hypertrophy markers (SOX9 and COL2) and reduced hypertrophy markers (COL10, MMP13, and ADAMTS4). Preconditioning with IGF1 has better and very significant results in lowering MMP13 and ADAMTS4 levels. The present study supports IGF1 pre-conditioned SMMSCs-CM to develop a new therapeutic approach in OA improvement through its chondrogenic and chondroprotective roles.
Keywords: Chondrogenesis; Col2; IGF1; MMP13; Mesenchymal Stem Cells.
© 2021 The Author(s).
Conflict of interest statement
The authors declare that there are no competing interests associated with the manuscript.
Figures
Similar articles
-
Anti‑inflammatory and anti‑catabolic effect of non‑animal stabilized hyaluronic acid and mesenchymal stem cell‑conditioned medium in an osteoarthritis coculture model.Mol Med Rep. 2020 May;21(5):2243-2250. doi: 10.3892/mmr.2020.11004. Epub 2020 Feb 26. Mol Med Rep. 2020. PMID: 32323772
-
MicroRNA-92a-3p Regulates Aggrecanase-1 and Aggrecanase-2 Expression in Chondrogenesis and IL-1β-Induced Catabolism in Human Articular Chondrocytes.Cell Physiol Biochem. 2017;44(1):38-52. doi: 10.1159/000484579. Epub 2017 Nov 3. Cell Physiol Biochem. 2017. PMID: 29241192
-
Mesenchymal stem cells secrete factors that inhibit inflammatory processes in short-term osteoarthritic synovium and cartilage explant culture.Osteoarthritis Cartilage. 2012 Oct;20(10):1186-96. doi: 10.1016/j.joca.2012.06.003. Epub 2012 Jul 5. Osteoarthritis Cartilage. 2012. PMID: 22771777
-
Recent advances in mesenchymal stem/stromal cells (MSCs)-based approaches for osteoarthritis (OA) therapy.Cell Biol Int. 2023 Jun;47(6):1033-1048. doi: 10.1002/cbin.12008. Epub 2023 Mar 30. Cell Biol Int. 2023. PMID: 36994843 Review.
-
Human umbilical cord mesenchymal stem cells promoting knee joint chondrogenesis for the treatment of knee osteoarthritis: a systematic review.J Orthop Surg Res. 2023 Aug 29;18(1):639. doi: 10.1186/s13018-023-04131-7. J Orthop Surg Res. 2023. PMID: 37644595 Free PMC article.
Cited by
-
Risk of cancer in acromegaly patients: An updated meta-analysis and systematic review.PLoS One. 2023 Nov 30;18(11):e0285335. doi: 10.1371/journal.pone.0285335. eCollection 2023. PLoS One. 2023. PMID: 38032888 Free PMC article.
-
The miR-130a-3p/TGF-βRII Axis Participates in Inhibiting the Differentiation of Fibroblasts Induced by TGF-β1.Front Pharmacol. 2021 Jul 28;12:732540. doi: 10.3389/fphar.2021.732540. eCollection 2021. Front Pharmacol. 2021. PMID: 34393805 Free PMC article.
-
Unraveling the complex interplay of sex, endocrinology, and inflammation in post-Injury articular cartilage breakdown through in silico modeling.Sci Rep. 2024 Nov 19;14(1):28654. doi: 10.1038/s41598-024-77730-x. Sci Rep. 2024. PMID: 39562596 Free PMC article.
-
Conditioned Medium - Is it an Undervalued Lab Waste with the Potential for Osteoarthritis Management?Stem Cell Rev Rep. 2023 Jul;19(5):1185-1213. doi: 10.1007/s12015-023-10517-1. Epub 2023 Feb 15. Stem Cell Rev Rep. 2023. PMID: 36790694 Free PMC article. Review.
-
The Characterization of Serum-Free Media on Human Mesenchymal Stem Cell Fibrochondrogenesis.Bioengineering (Basel). 2025 May 19;12(5):546. doi: 10.3390/bioengineering12050546. Bioengineering (Basel). 2025. PMID: 40428164 Free PMC article.
References
-
- Duval E., Leclercq S., Elissalde J.M., Demoor M., Galéra P. and Boumédiene K. (2009) Hypoxia-inducible factor 1α inhibits the fibroblast-like markers type I and type III collagen during hypoxia-induced chondrocyte redifferentiation: Hypoxia not only induces type II collagen and aggrecan, but it also inhibits type I and type III collagen in the hypoxia-inducible factor 1α-dependent redifferentiation of chondrocytes. Arthritis Rheum. 60, 3038–3048 10.1002/art.24851 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

