Functional development and regeneration of hair cells in the zebrafish lateral line
- PMID: 34143497
- PMCID: PMC7612129
- DOI: 10.1113/JP281522
Functional development and regeneration of hair cells in the zebrafish lateral line
Abstract
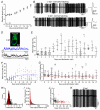
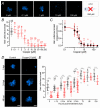
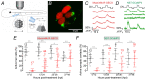
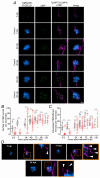
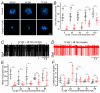
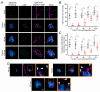
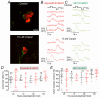
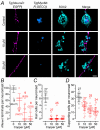
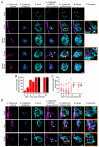
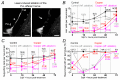
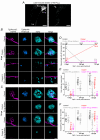
Key points: We investigated hair-cell regeneration in the zebrafish lateral line following the application of the ototoxic compound copper. In early-larval zebrafish (<10 days post-fertilisation), regenerated hair cells drive action potentials (APs) in the afferent neurons 24 h post-copper treatment (24 hpt). Full regeneration of the early-larval neuromasts, the organs containing the hair cells, requires ∼48 h due to the progressive addition of hair cells and synaptic refinement. In older larval zebrafish, regenerated hair cells are active and drive afferent APs by 48 hpt, which is comparable to larvae, but the functional recovery of their neuromasts requires >120 hpt. Afferent terminals within the regenerating neuromast appear to initially contact supporting cells, and their complete ablation prevents the timely reappearance of supporting cells and hair cells. We conclude that the regeneration of zebrafish neuromasts is slower after the initial developmental stages, and that the afferent input plays a key role in driving this process.
Abstract: Hair cells are mechanosensory receptors responsible for transducing auditory and vestibular information into electrical signals, which are then transmitted with remarkable precision to afferent neurons. Different from mammals, the hair cells of lower vertebrates, including those present in the neuromasts of the zebrafish lateral line, regenerate following environmental or chemical insults. Here we investigate the time course of regeneration of hair cells in vivo using electrophysiology, two-photon imaging and immunostaining applied to wild-type and genetically encoded fluorescent indicator zebrafish lines. Functional hair cells drive spontaneous action potentials in the posterior lateral line afferent fibres, the frequency of which progressively increases over the first 10 days post-fertilisation (dpf). Higher firing-rate fibres are only observed from ∼6 dpf. Following copper treatment, newly formed hair cells become functional and are able to drive APs in the afferent fibres within 48 h in both early-larval (≤8 dpf) and late-larval (12-17 dpf) zebrafish. However, the complete functional regeneration of the entire neuromast is delayed in late-larval compared to early-larval zebrafish. We propose that while individual regenerating hair cells can rapidly become active, the acquisition of fully functional neuromasts progresses faster at early-larval stages, a time when hair cells are still under development. At both ages, the afferent terminals in the regenerating neuromast appear to make initial contact with supporting cells. The ablation of the lateral line afferent neurons prevents the timely regeneration of supporting cells and hair cells. These findings indicate that the afferent system is likely to facilitate or promote the neuromast regeneration process.
Keywords: afferent fibres; development; electrophysiology; hair cells; imaging; neuromast; otoxic compound; regeneration; ribbon synapses; zebrafish.
© 2021 The Authors. The Journal of Physiology published by John Wiley & Sons Ltd on behalf of The Physiological Society.
Conflict of interest statement
Figures


Comment in
-
Tail of two fishies: age and afferents influence zebrafish lateral-line hair cell regeneration.J Physiol. 2021 Aug;599(16):3801-3802. doi: 10.1113/JP282012. Epub 2021 Jul 22. J Physiol. 2021. PMID: 34245018 Free PMC article. No abstract available.
Similar articles
-
Phoenix is required for mechanosensory hair cell regeneration in the zebrafish lateral line.PLoS Genet. 2009 Apr;5(4):e1000455. doi: 10.1371/journal.pgen.1000455. Epub 2009 Apr 17. PLoS Genet. 2009. PMID: 19381250 Free PMC article.
-
Mechanical overstimulation causes acute injury and synapse loss followed by fast recovery in lateral-line neuromasts of larval zebrafish.Elife. 2021 Oct 19;10:e69264. doi: 10.7554/eLife.69264. Elife. 2021. PMID: 34665127 Free PMC article.
-
Sub-lethal concentrations of waterborne copper are toxic to lateral line neuromasts in zebrafish (Danio rerio).Hear Res. 2006 Mar;213(1-2):1-10. doi: 10.1016/j.heares.2005.10.015. Epub 2005 Dec 28. Hear Res. 2006. PMID: 16386394
-
Sensory hair cell regeneration in the zebrafish lateral line.Dev Dyn. 2014 Oct;243(10):1187-202. doi: 10.1002/dvdy.24167. Epub 2014 Aug 14. Dev Dyn. 2014. PMID: 25045019 Free PMC article. Review.
-
The development of lateral line placodes: taking a broader view.Dev Biol. 2014 May 1;389(1):68-81. doi: 10.1016/j.ydbio.2014.02.016. Epub 2014 Feb 26. Dev Biol. 2014. PMID: 24582732 Review.
Cited by
-
Tail of two fishies: age and afferents influence zebrafish lateral-line hair cell regeneration.J Physiol. 2021 Aug;599(16):3801-3802. doi: 10.1113/JP282012. Epub 2021 Jul 22. J Physiol. 2021. PMID: 34245018 Free PMC article. No abstract available.
-
Tekt3 Safeguards Proper Functions and Morphology of Neuromast Hair Bundles.Int J Mol Sci. 2025 Mar 28;26(7):3115. doi: 10.3390/ijms26073115. Int J Mol Sci. 2025. PMID: 40243732 Free PMC article.
-
Modeling of Charcot-Marie-Tooth disease in zebrafish.Front Mol Neurosci. 2025 Aug 4;18:1641793. doi: 10.3389/fnmol.2025.1641793. eCollection 2025. Front Mol Neurosci. 2025. PMID: 40831607 Free PMC article. Review.
-
Vestibular physiology and function in zebrafish.Front Cell Dev Biol. 2023 Apr 18;11:1172933. doi: 10.3389/fcell.2023.1172933. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37143895 Free PMC article. Review.
-
The ndrg2 Gene Regulates Hair Cell Morphogenesis and Auditory Function during Zebrafish Development.Int J Mol Sci. 2023 Jun 11;24(12):10002. doi: 10.3390/ijms241210002. Int J Mol Sci. 2023. PMID: 37373150 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

