High-throughput screening and rational design of biofunctionalized surfaces with optimized biocompatibility and antimicrobial activity
- PMID: 34145249
- PMCID: PMC8213795
- DOI: 10.1038/s41467-021-23954-8
High-throughput screening and rational design of biofunctionalized surfaces with optimized biocompatibility and antimicrobial activity
Abstract
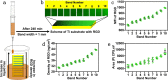
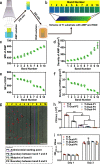
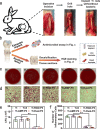
Peptides are widely used for surface modification to develop improved implants, such as cell adhesion RGD peptide and antimicrobial peptide (AMP). However, it is a daunting challenge to identify an optimized condition with the two peptides showing their intended activities and the parameters for reaching such a condition. Herein, we develop a high-throughput strategy, preparing titanium (Ti) surfaces with a gradient in peptide density by click reaction as a platform, to screen the positions with desired functions. Such positions are corresponding to optimized molecular parameters (peptide densities/ratios) and associated preparation parameters (reaction times/reactant concentrations). These parameters are then extracted to prepare nongradient mono- and dual-peptide functionalized Ti surfaces with desired biocompatibility or/and antimicrobial activity in vitro and in vivo. We also demonstrate this strategy could be extended to other materials. Here, we show that the high-throughput versatile strategy holds great promise for rational design and preparation of functional biomaterial surfaces.
Conflict of interest statement
L.W., Z.F., J.C., G.H., and H.X. have applied for patents related to this study. The remaining authors declare no competing interests.
Figures





Similar articles
-
Preparing and immobilizing antimicrobial osteogenic growth peptide on titanium substrate surface.J Biomed Mater Res A. 2018 Dec;106(12):3021-3033. doi: 10.1002/jbm.a.36491. Epub 2018 Sep 7. J Biomed Mater Res A. 2018. PMID: 30260556
-
Atomic layer deposition of nano-TiO2 thin films with enhanced biocompatibility and antimicrobial activity for orthopedic implants.Int J Nanomedicine. 2017 Dec 8;12:8711-8723. doi: 10.2147/IJN.S148065. eCollection 2017. Int J Nanomedicine. 2017. PMID: 29263665 Free PMC article.
-
Drug release and bone growth studies of antimicrobial peptide-loaded calcium phosphate coating on titanium.J Biomed Mater Res B Appl Biomater. 2012 Jul;100(5):1344-52. doi: 10.1002/jbm.b.32701. Epub 2012 May 7. J Biomed Mater Res B Appl Biomater. 2012. PMID: 22566395
-
[Progress in antibacterial/osteogenesis dual-functional surface modification strategy of titanium-based implants].Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2023 Oct 15;37(10):1300-1313. doi: 10.7507/1002-1892.202306025. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2023. PMID: 37848328 Free PMC article. Review. Chinese.
-
Review of titanium surface modification techniques and coatings for antibacterial applications.Acta Biomater. 2019 Jan 1;83:37-54. doi: 10.1016/j.actbio.2018.10.036. Epub 2018 Oct 26. Acta Biomater. 2019. PMID: 30541702 Review.
Cited by
-
Construction and high-throughput screening of gradient nanowire coatings on titanium surface towards ameliorated osseointegration.Mater Today Bio. 2024 Dec 7;30:101392. doi: 10.1016/j.mtbio.2024.101392. eCollection 2025 Feb. Mater Today Bio. 2024. PMID: 39759850 Free PMC article.
-
Near-infrared smart responsive orthopedic implants with synergistic antimicrobial and bone integration-promoting properties.J Orthop Translat. 2025 Apr 9;52:55-69. doi: 10.1016/j.jot.2025.03.015. eCollection 2025 May. J Orthop Translat. 2025. PMID: 40248127 Free PMC article.
-
The Potential of Surface-Immobilized Antimicrobial Peptides for the Enhancement of Orthopaedic Medical Devices: A Review.Antibiotics (Basel). 2023 Jan 19;12(2):211. doi: 10.3390/antibiotics12020211. Antibiotics (Basel). 2023. PMID: 36830122 Free PMC article. Review.
-
A paradigm for high-throughput screening of cell-selective surfaces coupling orthogonal gradients and machine learning-based cell recognition.Bioact Mater. 2023 May 5;28:1-11. doi: 10.1016/j.bioactmat.2023.04.022. eCollection 2023 Oct. Bioact Mater. 2023. PMID: 37214260 Free PMC article.
-
In Vivo Antibacterial Efficacy of Antimicrobial Peptides Modified Metallic Implants─Systematic Review and Meta-Analysis.ACS Biomater Sci Eng. 2022 May 9;8(5):1749-1762. doi: 10.1021/acsbiomaterials.1c01307. Epub 2022 Apr 12. ACS Biomater Sci Eng. 2022. PMID: 35412810 Free PMC article.
References
-
- Goddard JM, Hotchkiss JH. Polymer surface modification for the attachment of bioactive compounds. Prog. Polym. Sci. 2007;32:698–725. doi: 10.1016/j.progpolymsci.2007.04.002. - DOI
-
- Bauer S, Schmuki P, von der Mark K, Park J. Engineering biocompatible implant surfaces: part I: materials and surfaces. Prog. Mater. Sci. 2013;58:261–326. doi: 10.1016/j.pmatsci.2012.09.001. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

