Strategies for Poly(3-hydroxybutyrate) Production Using a Cold-Shock Promoter in Escherichia coli
- PMID: 34150730
- PMCID: PMC8211017
- DOI: 10.3389/fbioe.2021.666036
Strategies for Poly(3-hydroxybutyrate) Production Using a Cold-Shock Promoter in Escherichia coli
Abstract
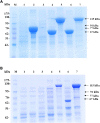
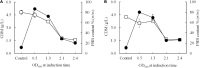
The present study attempted to increase poly(3-hydroxybutyrate) (PHB) production by improving expression of PHB biosynthesis operon derived from Cupriavidus necator strain A-04 using various types of promoters. The intact PHB biosynthesis operon of C. necator A-04, an alkaline tolerant strain isolated in Thailand with a high degree of 16S rRNA sequence similarity with C. necator H16, was subcloned into pGEX-6P-1, pColdI, pColdTF, pBAD/Thio-TOPO, and pUC19 (native promoter) and transformed into Escherichia coli JM109. While the phaC A-04 gene was insoluble in most expression systems tested, it became soluble when it was expressed as a fusion protein with trigger factor (TF), a ribosome associated bacterial chaperone, under the control of a cold shock promoter. Careful optimization indicates that the cold-shock cspA promoter enhanced phaCA-04 protein expression and the chaperone function of TF play critical roles in increasing soluble phaCA-04 protein. Induction strategies and parameters in flask experiments were optimized to obtain high expression of soluble PhaCA-04 protein with high YP/S and PHB productivity. Soluble phaCA-04 was purified through immobilized metal affinity chromatography (IMAC). The results demonstrated that the soluble phaCA-04 from pColdTF-phaCAB A-04 was expressed at a level of as high as 47.4 ± 2.4% of total protein and pColdTF-phaCAB A-04 enhanced soluble protein formation to approximately 3.09-4.1 times higher than that from pColdI-phaCAB A-04 by both conventional method and short induction method developed in this study. Cultivation in a 5-L fermenter led to PHB production of 89.8 ± 2.3% PHB content, a YP/S value of 0.38 g PHB/g glucose and a productivity of 0.43 g PHB/(L.h) using pColdTF-phaCAB A-04. The PHB film exhibited high optical transparency and possessed Mw 5.79 × 105 Da, Mn 1.86 × 105 Da, and PDI 3.11 with normal melting temperature and mechanical properties.
Keywords: Cupriavidus necator; E. coli – Escherichia coli; cold shock; cspA gene; pCold; polyhydroxybutyrate.
Copyright © 2021 Boontip, Waditee-Sirisattha, Honda and Napathorn.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





Similar articles
-
Polyhydroxybutyrate (PHB) Production Using an Arabinose-Inducible Expression System in Comparison With Cold Shock Inducible Expression System in Escherichia coli.Front Bioeng Biotechnol. 2021 May 3;9:661096. doi: 10.3389/fbioe.2021.661096. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 34012957 Free PMC article.
-
Enhanced polyhydroxybutyrate (PHB) production via the coexpressed phaCAB and vgb genes controlled by arabinose P promoter in Escherichia coli.Lett Appl Microbiol. 2010 Feb;50(2):158-67. doi: 10.1111/j.1472-765X.2009.02772.x. Epub 2009 Nov 12. Lett Appl Microbiol. 2010. PMID: 19943886
-
Involvement of the AtoSCDAEB regulon in the high molecular weight poly-(R)-3-hydroxybutyrate biosynthesis in phaCAB(+)Escherichia coli.Metab Eng. 2012 Jul;14(4):354-65. doi: 10.1016/j.ymben.2012.03.010. Epub 2012 Mar 30. Metab Eng. 2012. PMID: 22484344
-
Functional expression of phaCAB genes from Cupriavidus taiwanensis strain 184 in Escherichia coli for polyhydroxybutyrate production.Appl Biochem Biotechnol. 2010 Dec;162(8):2355-64. doi: 10.1007/s12010-010-9008-y. Epub 2010 Jun 16. Appl Biochem Biotechnol. 2010. PMID: 20556541
-
A Review on Enhancing Cupriavidus necator Fermentation for Poly(3-hydroxybutyrate) (PHB) Production From Low-Cost Carbon Sources.Front Bioeng Biotechnol. 2022 Jul 19;10:946085. doi: 10.3389/fbioe.2022.946085. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35928944 Free PMC article. Review.
Cited by
-
Random genome reduction coupled with polyhydroxybutyrate biosynthesis to facilitate its accumulation in Escherichia coli.Front Bioeng Biotechnol. 2022 Aug 29;10:978211. doi: 10.3389/fbioe.2022.978211. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36105609 Free PMC article.
-
Recent advances in engineering non-native microorganisms for poly(3-hydroxybutyrate) production.World J Microbiol Biotechnol. 2025 Jan 24;41(2):48. doi: 10.1007/s11274-025-04261-6. World J Microbiol Biotechnol. 2025. PMID: 39849243 Review.
-
Synthetic microbes and biocatalyst designs in Thailand.Biotechnol Notes. 2023 Feb 23;4:28-40. doi: 10.1016/j.biotno.2023.02.003. eCollection 2023. Biotechnol Notes. 2023. PMID: 39416912 Free PMC article. Review.
-
Construction of an Antibiotic-Free Vector and its Application in the Metabolic Engineering of Escherichia Coli for Polyhydroxybutyrate Production.Front Bioeng Biotechnol. 2022 Jun 3;10:837944. doi: 10.3389/fbioe.2022.837944. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 35721860 Free PMC article.
References
-
- Agus J., Kahar P., Abe H., Doi Y., Tsuge T. (2006a). Altered expression of polyhydroxyalkanoate synthase gene and its effect on poly [(R)-3-hydroxybutyrate] synthesis in recombinant Escherichia coli. Polym. Degrad. Stab. 91 1645–1650. 10.1016/j.polymdegradstab.2005.12.011 - DOI
-
- Agus J., Kahar P., Abe H., Doi Y., Tsuge T. (2006b). Molecular weight characterization of poly [(R)-3-hydroxybutyrate] synthesized by genetically engineered strains of Escherichia coli. Polym. Degrad. Stab. 91 1138–1146. 10.1016/j.polymdegradstab.2005.07.006 - DOI
-
- Baneyx F. (1999). Recombinant protein expression in Escherichia coli. Curr. Opin. Biotechnol. 10 411–421. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

