Identification of Quinolinones as Antivirals against Venezuelan Equine Encephalitis Virus
- PMID: 34152810
- PMCID: PMC8373297
- DOI: 10.1128/AAC.00244-21
Identification of Quinolinones as Antivirals against Venezuelan Equine Encephalitis Virus
Abstract
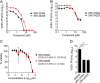
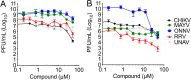
Venezuelan equine encephalitis virus (VEEV) is a reemerging alphavirus that can cause encephalitis resulting in severe human morbidity and mortality. Using a high-throughput cell-based screen, we identified a quinolinone compound that protected against VEEV-induced cytopathic effects. Analysis of viral replication in cells identified several quinolinone compounds with potent inhibitory activity against vaccine and virulent strains of VEEV. These quinolinones also displayed inhibitory activity against additional alphaviruses, such as Mayaro virus and Ross River virus, although the potency was greatly reduced. Time-of-addition studies indicated that these compounds inhibit the early-to-mid stage of viral replication. Deep sequencing and reverse genetics studies identified two unique resistance mutations in the nsP2 gene (Y102S/C; stalk domain) that conferred VEEV resistance on this chemical series. Moreover, introduction of a K102Y mutation into the nsP2 gene enhanced the sensitivity of chikungunya virus (CHIKV) to this chemical series. Computational modeling of CHIKV and VEEV nsP2 identified a highly probable docking alignment for the quinolinone compounds that require a tyrosine residue at position 102 within the helicase stalk domain. These studies identified a class of compounds with antiviral activity against VEEV and other alphaviruses and provide further evidence that therapeutics targeting nsP2 may be useful against alphavirus infection.
Keywords: alphavirus; antiviral agents.
Figures




Similar articles
-
Discovery of a novel compound with anti-venezuelan equine encephalitis virus activity that targets the nonstructural protein 2.PLoS Pathog. 2014 Jun 26;10(6):e1004213. doi: 10.1371/journal.ppat.1004213. eCollection 2014 Jun. PLoS Pathog. 2014. PMID: 24967809 Free PMC article.
-
Mutations in Hypervariable Domain of Venezuelan Equine Encephalitis Virus nsP3 Protein Differentially Affect Viral Replication.J Virol. 2020 Jan 17;94(3):e01841-19. doi: 10.1128/JVI.01841-19. Print 2020 Jan 17. J Virol. 2020. PMID: 31694937 Free PMC article.
-
Benzamidine ML336 inhibits plus and minus strand RNA synthesis of Venezuelan equine encephalitis virus without affecting host RNA production.Antiviral Res. 2020 Feb;174:104674. doi: 10.1016/j.antiviral.2019.104674. Epub 2019 Dec 6. Antiviral Res. 2020. PMID: 31816348 Free PMC article.
-
Current Understanding of the Molecular Basis of Venezuelan Equine Encephalitis Virus Pathogenesis and Vaccine Development.Viruses. 2019 Feb 18;11(2):164. doi: 10.3390/v11020164. Viruses. 2019. PMID: 30781656 Free PMC article. Review.
-
Venezuelan Equine Encephalitis Virus Capsid-The Clever Caper.Viruses. 2017 Sep 29;9(10):279. doi: 10.3390/v9100279. Viruses. 2017. PMID: 28961161 Free PMC article. Review.
Cited by
-
Advances in the Development of Small Molecule Antivirals against Equine Encephalitic Viruses.Viruses. 2023 Feb 1;15(2):413. doi: 10.3390/v15020413. Viruses. 2023. PMID: 36851628 Free PMC article. Review.
-
Third intracellular loop of HCMV US28 is necessary for signaling and viral reactivation.J Virol. 2025 Jan 31;99(1):e0180124. doi: 10.1128/jvi.01801-24. Epub 2024 Dec 10. J Virol. 2025. PMID: 39655954 Free PMC article.
-
Optimization of 4-Anilinoquinolines as Dengue Virus Inhibitors.Molecules. 2021 Dec 3;26(23):7338. doi: 10.3390/molecules26237338. Molecules. 2021. PMID: 34885921 Free PMC article.
-
A high-throughput response to the SARS-CoV-2 pandemic.SLAS Discov. 2024 Jul;29(5):100160. doi: 10.1016/j.slasd.2024.100160. Epub 2024 May 16. SLAS Discov. 2024. PMID: 38761981 Free PMC article.
-
Novel and repurposed antiviral molecules for arbovirus infections with epidemic Potential: A systematic review.New Microbes New Infect. 2025 Jul 10;66:101614. doi: 10.1016/j.nmni.2025.101614. eCollection 2025 Aug. New Microbes New Infect. 2025. PMID: 40776987 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

