C1q binding to surface-bound IgG is stabilized by C1r2s2 proteases
- PMID: 34155115
- PMCID: PMC8256010
- DOI: 10.1073/pnas.2102787118
C1q binding to surface-bound IgG is stabilized by C1r2s2 proteases
Abstract
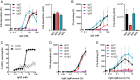
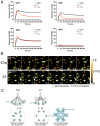
Complement is an important effector mechanism for antibody-mediated clearance of infections and tumor cells. Upon binding to target cells, the antibody's constant (Fc) domain recruits complement component C1 to initiate a proteolytic cascade that generates lytic pores and stimulates phagocytosis. The C1 complex (C1qr2s2) consists of the large recognition protein C1q and a heterotetramer of proteases C1r and C1s (C1r2s2). While interactions between C1 and IgG-Fc are believed to be mediated by the globular heads of C1q, we here find that C1r2s2 proteases affect the capacity of C1q to form an avid complex with surface-bound IgG molecules (on various 2,4-dinitrophenol [DNP]-coated surfaces and pathogenic Staphylococcus aureus). The extent to which C1r2s2 contributes to C1q-IgG stability strongly differs between human IgG subclasses. Using antibody engineering of monoclonal IgG, we reveal that hexamer-enhancing mutations improve C1q-IgG stability, both in the absence and presence of C1r2s2 In addition, hexamer-enhanced IgGs targeting S. aureus mediate improved complement-dependent phagocytosis by human neutrophils. Altogether, these molecular insights into complement binding to surface-bound IgGs could be important for optimal design of antibody therapies.
Keywords: C1; IgG hexamerization; IgG subclasses; Staphylococcus aureus; complement.
Copyright © 2021 the Author(s). Published by PNAS.
Conflict of interest statement
Competing interest statement: A.K., J.A.G.v.S., P.W.H.I.P., K.P.M.v.K., F.J.B., J. Schuurman, and S.H.M.R. are coinventors on a patent describing antibody therapies against S. aureus.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

