Early defects in mucopolysaccharidosis type IIIC disrupt excitatory synaptic transmission
- PMID: 34156977
- PMCID: PMC8410035
- DOI: 10.1172/jci.insight.142073
Early defects in mucopolysaccharidosis type IIIC disrupt excitatory synaptic transmission
Abstract
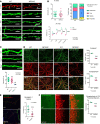
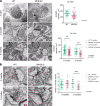
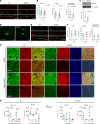
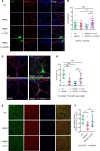
The majority of patients affected with lysosomal storage disorders (LSD) exhibit neurological symptoms. For mucopolysaccharidosis type IIIC (MPSIIIC), the major burdens are progressive and severe neuropsychiatric problems and dementia, primarily thought to stem from neurodegeneration. Using the MPSIIIC mouse model, we studied whether clinical manifestations preceding massive neurodegeneration arise from synaptic dysfunction. Reduced levels or abnormal distribution of multiple synaptic proteins were revealed in cultured hippocampal and CA1 pyramidal MPSIIIC neurons. These defects were rescued by virus-mediated gene correction. Dendritic spines were reduced in pyramidal neurons of mouse models of MPSIIIC and other (Tay-Sachs, sialidosis) LSD as early as at P10. MPSIIIC neurons also presented alterations in frequency and amplitude of miniature excitatory and inhibitory postsynaptic currents, sparse synaptic vesicles, reduced postsynaptic densities, disorganized microtubule networks, and partially impaired axonal transport of synaptic proteins. Furthermore, postsynaptic densities were reduced in postmortem cortices of human MPS patients, suggesting that the pathology is a common hallmark for neurological LSD. Together, our results demonstrate that lysosomal storage defects cause early alterations in synaptic structure and abnormalities in neurotransmission originating from impaired synaptic vesicular transport, and they suggest that synaptic defects could be targeted to treat behavioral and cognitive defects in neurological LSD patients.
Keywords: Genetics; Lysosomes; Neuroscience; Synapses.
Conflict of interest statement
Figures





References
-
- Scriver CR, et al. The Metabolic and Molecular Bases of Inherited Disease. McGraw-Hill Professional; 2001.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

