Real-world treatment patterns and disease control over one year in patients with inflammatory bowel disease in Brazil
- PMID: 34163120
- PMCID: PMC8218356
- DOI: 10.3748/wjg.v27.i23.3396
Real-world treatment patterns and disease control over one year in patients with inflammatory bowel disease in Brazil
Abstract
Background: Crohn's disease (CD) and ulcerative colitis (UC) are inflammatory bowel diseases (IBDs) with a remission-relapsing presentation and symptomatic exacerbations that have detrimental impacts on patient quality of life and are associated with a high cost burden, especially in patients with moderate-to-severe disease. The Real-world Data of Moderate-to-Severe Inflammatory Bowel Disease in Brazil (RISE BR) study was a noninterventional study designed to evaluate disease control, treatment patterns, disease burden and health-related quality of life in patients with moderate-to-severe active IBD. We report findings from the prospective follow-up phase of the RISE BR study in patients with active UC or CD.
Aim: To describe the 12-mo disease evolution and treatment patterns among patients with active moderate-to-severe IBD in Brazil.
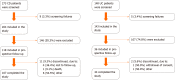
Methods: This was a prospective, noninterventional study of adult patients with active Crohn's disease (CD: Harvey-Bradshaw Index ≥ 8, CD Activity Index ≥ 220), inadequate CD control (i.e., calprotectin > 200 µg/g or colonoscopy previous results), or active ulcerative colitis (UC: Partial Mayo score ≥ 5). Enrollment occurred in 14 centers from October 2016 to February 2017. The proportion of active IBD patients after 9-12 mo of follow-up, Kaplan-Meier estimates of the time to mild or no activity and a summary of treatment initiation, discontinuation and dose changes were examined.
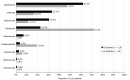
Results: The study included 118 CD and 36 UC patients, with mean ± SD ages of 43.3 ± 12.6 and 44.9 ± 16.5 years, respectively. The most frequent drug classes at index were biologics for CD (62.7%) and 5-aminosalicylate derivates for UC patients (91.7%). During follow-up, 65.3% of CD and 86.1% of UC patients initiated a new treatment at least once. Discontinuations/dose changes occurred in 68.1% of CD patients [median 2.0 (IQR: 2-5)] and 94.3% of UC patients [median 4.0 (IQR: 3-7)]. On average, CD and UC patients had 4.4 ± 2.6 and 5.0 ± 3.3 outpatient visits, respectively. The median time to first mild or no activity was 319 (IQR: 239-358) d for CD and 320 (IQR: 288-358) d for UC patients. At 9-12 mo, 22.0% of CD and 20.0% of UC patients had active disease.
Conclusion: Although a marked proportion of active IBD patients achieved disease control within one year, the considerable time to achieve this outcome represents an unmet medical need of the current standard of care in a Brazilian real-world setting.
Keywords: Crohn’s disease; Inflammatory bowel diseases; Prospective study; Ulcerative colitis.
©The Author(s) 2021. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: Ligia Yukie Sassaki has received fees for serving as a speaker for AbbVie and Takeda. Sender Jankiel Miszputen has received fees for serving as a speaker and/or a consultant for Farmoquimica, Janssen and Marjan. He has received research funding from Ache, Roche and Takeda. Wilson Roberto Catapani has received fees for serving as a speaker and/or an advisory board member for Janssen and Takeda. Mauro Bafutto has received fees for serving as a speaker for Takeda, AbbVie, Janssen, UCB and Farmoquimica. He has received fees for serving as an advisory board member for AbbVie and Janssen. Antonio Scafuto Scotton has received fees for serving as a speaker for Janssen, Novartis, AbbVie, MSD, EMS. He has received research funding from Janssen, Novartis, AbbVie, Roche, Pfizer, Bristol, Lilly, Novo Nordisk, Anthera, AstraZeneca, GSK, UCB, Sanofi, Takeda, Parexel, IQVIA, PPD, PRA, ICON, INP Research, Covance, In Trials. Cyrla Zaltman has received fees for serving as a speaker for UCB, Janssen, Takeda, AbbVie. She has received research funding from AbbVie, Takeda, Janssen. Julio Pinheiro Baima has received fees for serving as a speaker for Janssen and Takeda. Cristina Flores has received fees for serving as a speaker for Janssen, Takeda, AbbVie. She has received fees for serving as an advisory board member for Janssen. Rodrigo Bremer Nones has received fees for serving as a speaker for AbbVie, Ferring Pharmaceuticals, Janssen, Nestle, Novartis, Pfizer, UCB Pharma and Takeda. Jose Miguel Luz Parente has received fees for serving as a speaker for Takeda. Julio Maria Fonseca Chebli has received fees for serving as a speaker for AbbVie, Janssen, UCB Pharma and Takeda. Maria de Lourdes de Abreu Ferrari has received fees for serving as a speaker and/or advisory board member for AbbVie, Ferring Pharmaceuticals, Janssen, UCB Pharma, and Takeda. Rogerio Serafim Parra has received fees for serving as a speaker and/or an advisory board member for AbbVie, Ferring Pharmaceuticals, Janssen, UCB Pharma and Takeda. Jose Joaquim Ribeiro da Rocha has received fees for serving as a speaker for Nestle. Marley Ribeiro Feitosa has received fees for serving as a speaker for AbbVie and Janssen. Rosana Fusaro Caratin was an employee of Takeda Pharmaceuticals Brazil at the time the manuscript was developed. Juliana Tosta Senra is an employee of Takeda Pharmaceuticals Brazil. Genoile Oliveira Santana has received fees for serving as a speaker for Takeda, AbbVie, Janssen, UCB Pharma. She has received research funding from Celgene and Roche. She has received fees for serving as an advisory board member for Janssen. No conflict of interest: Roberto Luiz Kaiser Junior, Mikaell Alexandre Gouvea Faria, Carolina Dias Goncalves, Isabella de Miranda Guimaraes, Heda Maria Barska dos Santos Amarante, Murilo Moura Lima, Odery Ramos, Omar Feres, Hagata Souza Ramos, Julia Faria Campos, and Maria das Gracas Pimenta Sanna.
Figures


References
-
- Mowat C, Cole A, Windsor A, Ahmad T, Arnott I, Driscoll R, Mitton S, Orchard T, Rutter M, Younge L, Lees C, Ho GT, Satsangi J, Bloom S IBD Section of the British Society of Gastroenterology. Guidelines for the management of inflammatory bowel disease in adults. Gut. 2011;60:571–607. - PubMed
-
- Gomollón F, Dignass A, Annese V, Tilg H, Van Assche G, Lindsay JO, Peyrin-Biroulet L, Cullen GJ, Daperno M, Kucharzik T, Rieder F, Almer S, Armuzzi A, Harbord M, Langhorst J, Sans M, Chowers Y, Fiorino G, Juillerat P, Mantzaris GJ, Rizzello F, Vavricka S, Gionchetti P ECCO. 3rd European Evidence-based Consensus on the Diagnosis and Management of Crohn's Disease 2016: Part 1: Diagnosis and Medical Management. J Crohns Colitis. 2017;11:3–25. - PubMed
-
- Brady JE, Stott-Miller M, Mu G, Perera S. Treatment Patterns and Sequencing in Patients With Inflammatory Bowel Disease. Clin Ther 2018; 40: 1509-1521. :e5. - PubMed
-
- Lichtenstein GR, Loftus EV, Isaacs KL, Regueiro MD, Gerson LB, Sands BE. ACG Clinical Guideline: Management of Crohn's Disease in Adults. Am J Gastroenterol. 2018;113:481–517. - PubMed
-
- Kornbluth A, Sachar DB Practice Parameters Committee of the American College of Gastroenterology. Ulcerative colitis practice guidelines in adults: American College Of Gastroenterology, Practice Parameters Committee. Am J Gastroenterol. 2010;105:501–23; quiz 524. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

