A novel catalytic heme cofactor in SfmD with a single thioether bond and a bis-His ligand set revealed by a de novo crystal structural and spectroscopic study
- PMID: 34163669
- PMCID: PMC8179489
- DOI: 10.1039/d0sc06369j
A novel catalytic heme cofactor in SfmD with a single thioether bond and a bis-His ligand set revealed by a de novo crystal structural and spectroscopic study
Abstract
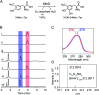
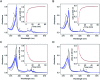
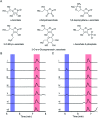
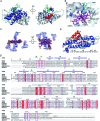
SfmD is a heme-dependent enzyme in the biosynthetic pathway of saframycin A. Here, we present a 1.78 Å resolution de novo crystal structure of SfmD, which unveils a novel heme cofactor attached to the protein with an unusual Hx n HxxxC motif (n ∼ 38). This heme cofactor is unique in two respects. It contains a single thioether bond in a cysteine-vinyl link with Cys317, and the ferric heme has two axial protein ligands, i.e., His274 and His313. We demonstrated that SfmD heme is catalytically active and can utilize dioxygen and ascorbate for a single-oxygen insertion into 3-methyl-l-tyrosine. Catalytic assays using ascorbate derivatives revealed the functional groups of ascorbate essential to its function as a cosubstrate. Abolishing the thioether linkage through mutation of Cys317 resulted in catalytically inactive SfmD variants. EPR and optical data revealed that the heme center undergoes a substantial conformational change with one axial histidine ligand dissociating from the iron ion in response to substrate 3-methyl-l-tyrosine binding or chemical reduction by a reducing agent, such as the cosubstrate ascorbate. The labile axial ligand was identified as His274 through redox-linked structural determinations. Together, identifying an unusual heme cofactor with a previously unknown heme-binding motif for a monooxygenase activity and the structural similarity of SfmD to the members of the heme-based tryptophan dioxygenase superfamily will broaden understanding of heme chemistry.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


Similar articles
-
Characterization of SfmD as a Heme peroxidase that catalyzes the regioselective hydroxylation of 3-methyltyrosine to 3-hydroxy-5-methyltyrosine in saframycin A biosynthesis.J Biol Chem. 2012 Feb 10;287(7):5112-21. doi: 10.1074/jbc.M111.306316. Epub 2011 Dec 20. J Biol Chem. 2012. PMID: 22187429 Free PMC article.
-
A new regime of heme-dependent aromatic oxygenase superfamily.Proc Natl Acad Sci U S A. 2021 Oct 26;118(43):e2106561118. doi: 10.1073/pnas.2106561118. Proc Natl Acad Sci U S A. 2021. PMID: 34667125 Free PMC article.
-
A possible role for the covalent heme-protein linkage in cytochrome c revealed via comparison of N-acetylmicroperoxidase-8 and a synthetic, monohistidine-coordinated heme peptide.Biochemistry. 2004 Feb 17;43(6):1656-66. doi: 10.1021/bi035531p. Biochemistry. 2004. PMID: 14769043
-
Different Mechanisms of Catalytic Complex Formation in Two L-Tryptophan Processing Dioxygenases.Front Mol Biosci. 2018 Jan 4;4:94. doi: 10.3389/fmolb.2017.00094. eCollection 2017. Front Mol Biosci. 2018. PMID: 29354636 Free PMC article. Review.
-
Ligand-protein interactions in nitric oxide synthase.J Inorg Biochem. 2005 Jan;99(1):306-23. doi: 10.1016/j.jinorgbio.2004.11.007. J Inorg Biochem. 2005. PMID: 15598509 Review.
Cited by
-
Structural insights into 2-oxindole-forming monooxygenase MarE: Divergent architecture and substrate positioning versus tryptophan dioxygenases.J Biol Chem. 2025 Mar;301(3):108241. doi: 10.1016/j.jbc.2025.108241. Epub 2025 Jan 27. J Biol Chem. 2025. PMID: 39880093 Free PMC article.
-
Combined spectroscopic and structural approaches to explore the mechanism of histidine-ligated heme-dependent aromatic oxygenases.Methods Enzymol. 2023;685:405-432. doi: 10.1016/bs.mie.2023.03.008. Epub 2023 May 2. Methods Enzymol. 2023. PMID: 37245909 Free PMC article.
-
Deciphering Tryptophan Oxygenation: Key Modulators of 2-Oxindole Formation in MarE.Angew Chem Int Ed Engl. 2025 Aug 25;64(35):e202510848. doi: 10.1002/anie.202510848. Epub 2025 Jul 20. Angew Chem Int Ed Engl. 2025. PMID: 40555689 Free PMC article.
-
Protein-derived cofactors: chemical innovations expanding enzyme catalysis.Chem Soc Rev. 2025 May 6;54(9):4502-4530. doi: 10.1039/d4cs00981a. Chem Soc Rev. 2025. PMID: 40151987 Free PMC article. Review.
-
Molecular Rationale for Partitioning between C-H and C-F Bond Activation in Heme-Dependent Tyrosine Hydroxylase.J Am Chem Soc. 2021 Mar 31;143(12):4680-4693. doi: 10.1021/jacs.1c00175. Epub 2021 Mar 18. J Am Chem Soc. 2021. PMID: 33734681 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

