Recent development in transition metal-catalysed C-H olefination
- PMID: 34164039
- PMCID: PMC8179420
- DOI: 10.1039/d0sc05555g
Recent development in transition metal-catalysed C-H olefination
Abstract
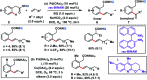
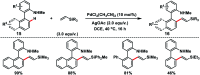
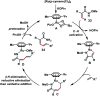
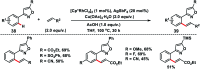
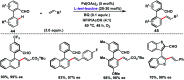
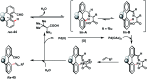
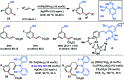
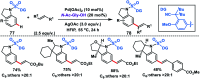
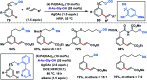
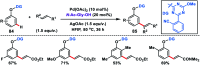
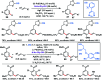
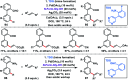
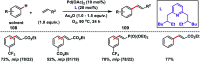
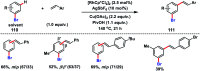
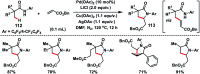
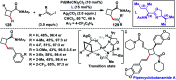
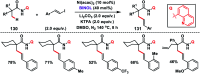
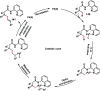
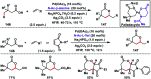
Transition metal-catalysed functionalizations of inert C-H bonds to construct C-C bonds represent an ideal route in the synthesis of valuable organic molecules. Fine tuning of directing groups, catalysts and ligands has played a crucial role in selective C-H bond (sp2 or sp3) activation. Recent developments in these areas have assured a high level of regioselectivity in C-H olefination reactions. In this review, we have summarized the recent progress in the oxidative olefination of sp2 and sp3 C-H bonds with special emphasis on distal, atroposelective, non-directed sp2 and directed sp3 C-H olefination. The scope, limitation, and mechanism of various transition metal-catalysed olefination reactions have been described briefly.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures








































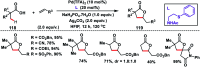














Similar articles
-
Emergence of Pyrimidine-Based meta-Directing Group: Journey from Weak to Strong Coordination in Diversifying meta-C-H Functionalization.Acc Chem Res. 2022 Feb 1;55(3):354-372. doi: 10.1021/acs.accounts.1c00629. Epub 2022 Jan 12. Acc Chem Res. 2022. PMID: 35021007
-
C-F and C-H bond activation of fluorobenzenes and fluoropyridines at transition metal centers: how fluorine tips the scales.Acc Chem Res. 2011 May 17;44(5):333-48. doi: 10.1021/ar100136x. Epub 2011 Mar 16. Acc Chem Res. 2011. PMID: 21410234
-
Diverse strategies for transition metal catalyzed distal C(sp3)-H functionalizations.Chem Sci. 2020 Sep 28;11(40):10887-10909. doi: 10.1039/d0sc04676k. Chem Sci. 2020. PMID: 34094339 Free PMC article. Review.
-
Photoredox Catalysis for Building C-C Bonds from C(sp2)-H Bonds.Chem Rev. 2018 Aug 22;118(16):7532-7585. doi: 10.1021/acs.chemrev.8b00077. Epub 2018 Jul 16. Chem Rev. 2018. PMID: 30011194
-
Transition metal catalysed direct construction of 2-pyridone scaffolds through C-H bond functionalizations.Org Biomol Chem. 2021 Dec 15;19(48):10516-10529. doi: 10.1039/d1ob01856f. Org Biomol Chem. 2021. PMID: 34816862 Review.
Cited by
-
Simplifying the Synthesis of Nonproteinogenic Amino Acids via Palladium-Catalyzed δ-Methyl C-H Olefination of Aliphatic Amines and Amino Acids.JACS Au. 2023 Jun 22;3(7):1975-1983. doi: 10.1021/jacsau.3c00215. eCollection 2023 Jul 24. JACS Au. 2023. PMID: 37502162 Free PMC article.
-
A Fujiwara-Moritani-Type Alkenylation Using a Traceless Directing Group Strategy: A Rare Example of C-C Bond Formation towards the C2-Carbon of Terminal Alkenes.European J Org Chem. 2023 Feb 17;26(8):e202201179. doi: 10.1002/ejoc.202201179. Epub 2023 Jan 26. European J Org Chem. 2023. PMID: 38504820 Free PMC article.
-
Non-directed Pd-catalysed electrooxidative olefination of arenes.Chem Sci. 2022 Aug 5;13(32):9432-9439. doi: 10.1039/d2sc03288k. eCollection 2022 Aug 17. Chem Sci. 2022. PMID: 36093017 Free PMC article.
-
Directing group assisted para-selective C-H alkynylation of unbiased arenes enabled by rhodium catalysis.Chem Sci. 2023 Sep 20;14(41):11381-11388. doi: 10.1039/d3sc03528j. eCollection 2023 Oct 25. Chem Sci. 2023. PMID: 37886091 Free PMC article.
-
Ru(ii)-catalyzed regioselective oxidative Heck reaction with internal olefins that tolerated strongly coordinating heterocycles.Chem Sci. 2024 Nov 19;15(47):20064-20072. doi: 10.1039/d4sc07036d. eCollection 2024 Dec 4. Chem Sci. 2024. PMID: 39568925 Free PMC article.
References
-
- McMurray L. O'Hara F. Gaunt M. J. Chem. Soc. Rev. 2011;40:1885–1898. - PubMed
- Fox J. C. Gilligan R. E. Pitts A. K. Bennett H. R. Gaunt M. J. Chem. Sci. 2016;7:2706–2710. - PMC - PubMed
- Hartwig J. F. J. Am. Chem. Soc. 2016;138:2–24. - PMC - PubMed
- Cernak T. Dykstra K. D. Tyagarajan S. Vachal P. Krska S. W. Chem. Soc. Rev. 2016;45:546–576. - PubMed
- Blakemore D. C. Castro L. Churcher I. Rees D. C. Thomas A. W. Wilson D. M. Wood A. Nat. Chem. 2018;10:383–394. - PubMed
-
- Ma W. Gandeepan P. Lid J. Ackermann L. Org. Chem. Front. 2017;4:1435–1467.
- Kozhushkov S. I. Ackermann L. Chem. Sci. 2013;4:886–896.
- Wedi P. van Gemmeren M. Angew. Chem., Int. Ed. 2018;57:13016–13027. - PubMed
- Dey A. Sinha S. K. Achar T. K. Maiti D. Angew. Chem., Int. Ed. 2019;58:10820–10843. - PubMed
- Deb A. Maiti D. Eur. J. Org. Chem. 2017:1239–1252.
- Bag S. Maiti D. Synthesis. 2016;48:804–815.
- Manikandana R. Jeganmohan M. Chem. Commun. 2017;53:8931–8947. - PubMed
- Rej S. Ano Y. Chatani N. Chem. Rev. 2020;120:1788–1887. - PubMed
-
- Wencel-Delord J. Glorius F. Nat. Chem. 2013;5:369–375. - PubMed
- Grimsdale A. C. Leok Chan K. Martin R. E. Jokisz P. G. Holmes A. B. Chem. Rev. 2009;109:897–1091. - PubMed
- Silva Paula M. M. Franco C. V. Baldin M. C. Rodrigues L. Barichello T. Savi G. D. Bellato L. F. Fiori M. A. Silva L. Mater. Sci. Eng., C. 2009;29:647–650.
Publication types
LinkOut - more resources
Full Text Sources

