Association of furosemide or hydrochlorothiazide use with risk of atrial fibrillation post pacemaker implantation among elderly patients
- PMID: 34164489
- PMCID: PMC8184456
- DOI: 10.21037/atm-21-1792
Association of furosemide or hydrochlorothiazide use with risk of atrial fibrillation post pacemaker implantation among elderly patients
Abstract
Background: Atrial fibrillation (AF) induced by artificial pacing is directly related to atrial remodeling. Previous basic research has shown that furosemide aggravates pathologic myocardial remodeling while hydrochlorothiazide alleviates it. However, whether furosemide or hydrochlorothiazide plays a role in developing AF after pacemaker implantation remains unknown. The study aims to investigate the association between oral furosemide or hydrochlorothiazide and the risk of developing AF after pacemaker implantation.
Methods: After a review of electronic medical records, elderly patients with pacemaker implantation and without a known baseline history of AF were included and information on their use of daily oral furosemide or hydrochlorothiazide was extracted. New incident AF cases were confirmed via the records of outpatient visits. A Cox proportional-hazards model was used to evaluate the association between daily oral furosemide or hydrochlorothiazide and risk of developing AF after pacemaker implantation, after adjustment for potential confounders.
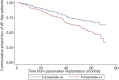
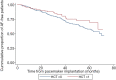
Results: Among a total of 551 patients aged more than 65 years, 157 AF cases were identified after pacemaker implantation during a maximum follow up of 3.0±1.6 years. Of these, 242 had used furosemide and 97 had used hydrochlorothiazide therapy. Patients taking daily oral furosemide had a relatively higher risk of AF after pacemaker implantation [hazard ratio (HR): 1.507, 95% confidence interval (CI): 1.036-2.192; P=0.032] after being adjusted for related disease and prescribed medications, while oral taking of hydrochlorothiazide was shown to be a non-effective factor (HR: 0.666, 95% CI: 0.413-1.074), which had no statistical significance.
Conclusions: Daily oral furosemide might increase the risk of developing AF after pacemaker implantation in elderly patients, while hydrochlorothiazide has no detrimental effect.
Keywords: Furosemide; atrial fibrillation (AF); elderly patients; hydrochlorothiazide; pacemaker implantation.
2021 Annals of Translational Medicine. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/atm-21-1792). The authors have no conflicts of interest to declare.
Figures
Similar articles
-
Association of angiotensin-converting enzyme inhibitors or angiotensin II receptor antagonists use with risk of atrial fibrillation after pacemaker implantation among very old patients.Biosci Trends. 2022 Jan 23;15(6):390-396. doi: 10.5582/bst.2021.01461. Epub 2021 Dec 4. Biosci Trends. 2022. PMID: 34866125
-
Predictors and clinical impact of atrial fibrillation after pacemaker implantation in elderly patients treated with dual chamber versus ventricular pacing.Pacing Clin Electrophysiol. 2003 Oct;26(10):2000-7. doi: 10.1046/j.1460-9592.2003.00309.x. Pacing Clin Electrophysiol. 2003. PMID: 14516342 Clinical Trial.
-
Analyses of risk factors and prognosis for new-onset atrial fibrillation in elderly patients after dual-chamber pacemaker implantation.J Geriatr Cardiol. 2018 Oct;15(10):628-633. doi: 10.11909/j.issn.1671-5411.2018.10.008. J Geriatr Cardiol. 2018. PMID: 30416511 Free PMC article.
-
Implication of ventricular pacing burden and atrial pacing therapies on the progression of atrial fibrillation: A systematic review and meta-analysis of randomized controlled trials.Heart Rhythm. 2019 Aug;16(8):1204-1214. doi: 10.1016/j.hrthm.2019.02.020. Epub 2019 Feb 14. Heart Rhythm. 2019. PMID: 30772532
-
Atrial pacing for the prevention and termination of atrial fibrillation.Am J Geriatr Cardiol. 2002 Nov-Dec;11(6):380-98. doi: 10.1111/j.1076-7460.2002.00072.x. Am J Geriatr Cardiol. 2002. PMID: 12417845 Review.
Cited by
-
Effects of ACEI/ARB or CCB use on atrial fibrillation in hypertensive patients following permanent pacemaker implantation.Front Cardiovasc Med. 2023 Jun 22;10:1191539. doi: 10.3389/fcvm.2023.1191539. eCollection 2023. Front Cardiovasc Med. 2023. PMID: 37424921 Free PMC article.
-
Drug-induced AF: Arrhythmogenic Mechanisms and Management Strategies.Arrhythm Electrophysiol Rev. 2024 Apr 3;13:e06. doi: 10.15420/aer.2023.24. eCollection 2024. Arrhythm Electrophysiol Rev. 2024. PMID: 38706787 Free PMC article. Review.
References
-
- Gregoratos G, Abrams J, Epstein AE, et al. ACC/AHA/NASPE 2002 guideline update for implantation of cardiac pacemakers and antiarrhythmia devices: summary article. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/NASPE Committee to Update the 1998 Pacemaker Guidelines). J Cardiovasc Electrophysiol 2002;13:1183-99. 10.1046/j.1540-8167.2002.01183.x - DOI - PubMed
-
- Nikolaidou T, Fox DJ, Brown BD. Bradycardia pacing. Medicine 2018;46:646-51. 10.1016/j.mpmed.2018.07.002 - DOI
-
- Sweeney MO, Hellkamp AS, Ellenbogen KA, et al. Adverse effect of ventricular pacing on heart failure and atrial fibrillation among patients with normal baseline QRS duration in a clinical trial of pacemaker therapy for sinus node dysfunction. Circulation 2003;107:2932-7. 10.1161/01.CIR.0000072769.17295.B1 - DOI - PubMed
LinkOut - more resources
Full Text Sources