Pathogenesis of human cytomegalovirus in the immunocompromised host
- PMID: 34168328
- PMCID: PMC8223196
- DOI: 10.1038/s41579-021-00582-z
Pathogenesis of human cytomegalovirus in the immunocompromised host
Abstract
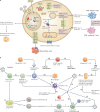
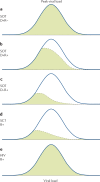
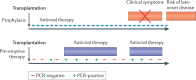
Human cytomegalovirus (HCMV) is a herpesvirus that infects ~60% of adults in developed countries and more than 90% in developing countries. Usually, it is controlled by a vigorous immune response so that infections are asymptomatic or symptoms are mild. However, if the immune system is compromised, HCMV can replicate to high levels and cause serious end organ disease. Substantial progress is being made in understanding the natural history and pathogenesis of HCMV infection and disease in the immunocompromised host. Serial measures of viral load defined the dynamics of HCMV replication and are now used routinely to allow intervention with antiviral drugs in individual patients. They are also used as pharmacodynamic read-outs to evaluate prototype vaccines that may protect against HCMV replication and to define immune correlates of this protection. This novel information is informing the design of randomized controlled trials of new antiviral drugs and vaccines currently under evaluation. In this Review, we discuss immune responses to HCMV and countermeasures deployed by the virus, the establishment of latency and reactivation from it, exogenous reinfection with additional strains, pathogenesis, development of end organ disease, indirect effects of infection, immune correlates of control of replication, current treatment strategies and the evaluation of novel vaccine candidates.
© 2021. Springer Nature Limited.
Conflict of interest statement
Both authors are co-inventors (along with I. Baraniak) on UK patent application number 2020135.6 assigned to University College London (UCL), entitled ‘hCMV antibody and vaccine target’, that deals with a novel antigenic domain on HCMV glycoprotein B (gB). UCL received funds from Takeda pharmaceuticals to compensate for the time P.G. spent as a member of the end-point committee for a randomized clinical trial (RCT) of maribavir. The authors declare no other competing interests.
Figures


References
-
- Zuhair M, et al. Estimation of the worldwide seroprevalence of cytomegalovirus: a systematic review and meta-analysis. Rev. Med. Virol. 2019;29:e2034. - PubMed
-
- Staras SA, et al. Cytomegalovirus seroprevalence and childhood sources of infection: a population-based study among pre-adolescents in the United States. J. Clin. Virol. 2008;43:266–271. - PubMed
-
- Griffiths PD, McLean A, Emery VC. Encouraging prospects for immunisation against primary cytomegalovirus infection. Vaccine. 2001;19:1356–1362. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

