The Innate Immune Glycoprotein Lactoferrin Represses the Helicobacter pylori cag Type IV Secretion System
- PMID: 34169626
- PMCID: PMC8560179
- DOI: 10.1002/cbic.202100249
The Innate Immune Glycoprotein Lactoferrin Represses the Helicobacter pylori cag Type IV Secretion System
Abstract
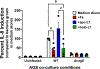
Chronic infection with Helicobacter pylori increases risk of gastric diseases including gastric cancer. Despite development of a robust immune response, H. pylori persists in the gastric niche. Progression of gastric inflammation to serious disease outcomes is associated with infection with H. pylori strains which encode the cag Type IV Secretion System (cag T4SS). The cag T4SS is responsible for translocating the oncogenic protein CagA into host cells and inducing pro-inflammatory and carcinogenic signaling cascades. Our previous work demonstrated that nutrient iron modulates the activity of the T4SS and biogenesis of T4SS pili. In response to H. pylori infection, the host produces a variety of antimicrobial molecules, including the iron-binding glycoprotein, lactoferrin. Our work shows that apo-lactoferrin exerts antimicrobial activity against H. pylori under iron-limited conditions, while holo-lactoferrin enhances bacterial growth. Culturing H. pylori in the presence of holo-lactoferrin prior to co-culture with gastric epithelial cells, results in repression of the cag T4SS activity. Concomitantly, a decrease in biogenesis of cag T4SS pili at the host-pathogen interface was observed under these culture conditions by high-resolution electron microscopy analyses. Taken together, these results indicate that acquisition of alternate sources of nutrient iron plays a role in regulating the pro-inflammatory activity of a bacterial secretion system and present novel therapeutic targets for the treatment of H. pylori-related disease.
Keywords: Helicobacter pylori; antimicrobial; bacterial pathogenesis; glycobiology; host-pathogen interactions; innate immunity; iron homeostasis; lactoferrin; toxin secretion; virulence.
© 2021 Wiley-VCH GmbH.
Figures


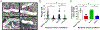

Similar articles
-
High resolution electron microscopy of the Helicobacter pylori Cag type IV secretion system pili produced in varying conditions of iron availability.J Vis Exp. 2014 Nov 21;(93):e52122. doi: 10.3791/52122. J Vis Exp. 2014. PMID: 25489938 Free PMC article.
-
Helicobacter pylori CagA and Cag type IV secretion system activity have key roles in triggering gastric transcriptional and proteomic alterations.Infect Immun. 2025 Apr 8;93(4):e0059524. doi: 10.1128/iai.00595-24. Epub 2025 Mar 6. Infect Immun. 2025. PMID: 40047510 Free PMC article.
-
TIFA Signaling in Gastric Epithelial Cells Initiates the cag Type 4 Secretion System-Dependent Innate Immune Response to Helicobacter pylori Infection.mBio. 2017 Aug 15;8(4):e01168-17. doi: 10.1128/mBio.01168-17. mBio. 2017. PMID: 28811347 Free PMC article.
-
The type IV secretion system in Helicobacter pylori.Future Microbiol. 2018 Jul;13:1041-1054. doi: 10.2217/fmb-2018-0038. Epub 2018 Jun 21. Future Microbiol. 2018. PMID: 29927340 Review.
-
Composition, structure and function of the Helicobacter pylori cag pathogenicity island encoded type IV secretion system.Future Microbiol. 2015;10(6):955-65. doi: 10.2217/fmb.15.32. Future Microbiol. 2015. PMID: 26059619 Free PMC article. Review.
Cited by
-
Lactotransferrin Downregulation Serves as a Potential Predictor for the Therapeutic Effectiveness of mTOR Inhibitors in the Metastatic Clear Cell Renal Cell Carcinoma without PTEN Mutation.Biomedicines. 2021 Dec 13;9(12):1896. doi: 10.3390/biomedicines9121896. Biomedicines. 2021. PMID: 34944711 Free PMC article.
-
Challenges and Prospects for Eradication of Helicobacter pylori: Targeting Virulence Factors, Metabolism, and Vaccine Innovation.Pathogens. 2025 Jun 21;14(7):619. doi: 10.3390/pathogens14070619. Pathogens. 2025. PMID: 40732667 Free PMC article. Review.
-
Antibiofilm Activity of Human Milk Oligosaccharides against Multidrug Resistant and Susceptible Isolates of Acinetobacter baumannii.ACS Infect Dis. 2021 Dec 10;7(12):3254-3263. doi: 10.1021/acsinfecdis.1c00420. Epub 2021 Nov 23. ACS Infect Dis. 2021. PMID: 34812035 Free PMC article.
-
Antimicrobial Effects of Lactoferrin against Helicobacter pylori Infection.Pathogens. 2023 Apr 14;12(4):599. doi: 10.3390/pathogens12040599. Pathogens. 2023. PMID: 37111484 Free PMC article. Review.
-
Helicobacter pylori infection in humans and phytotherapy, probiotics, and emerging therapeutic interventions: a review.Front Microbiol. 2024 Jan 10;14:1330029. doi: 10.3389/fmicb.2023.1330029. eCollection 2023. Front Microbiol. 2024. PMID: 38268702 Free PMC article. Review.
References
-
- Anderson BF, Baker HM, Norris GE, Rice DW, Baker EN, J. Mol. Biol. 1989, 209, Structure of human lactoferrin: Crystallographic s. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- UL1 TR000445/TR/NCATS NIH HHS/United States
- Office of Medical Research
- 1400969/National Science Foundation
- Core Scholarship
- IK2BX001701/Career Development
- T32 GM139800/GM/NIGMS NIH HHS/United States
- 1547757/National Science Foundation
- P01 CA116087/CA/NCI NIH HHS/United States
- Tissue Morphology Subcore
- Vanderbilt Institute for Clinical and Translational Research
- UL1 RR024975/RR/NCRR NIH HHS/United States
- UL1 TR002243/TR/NCATS NIH HHS/United States
- K08 AI151100/AI/NIAID NIH HHS/United States
- T32-AI095202/Childhood Infections Research
- R35 GM133602/GM/NIGMS NIH HHS/United States
- IK2 BX001701/BX/BLRD VA/United States
- T32 AI095202/AI/NIAID NIH HHS/United States
- T32 HL007411-36S1/GF/NIH HHS/United States
- R01 HD090061/HD/NICHD NIH HHS/United States
- R35 GM133602/Childhood Infections Research
- P30 DK058404/DK/NIDDK NIH HHS/United States
- VA/VA/United States
- Vanderbilt University Medical Center's Digestive Disease Research Center
- 5UL1TR002243-03/TR/NCATS NIH HHS/United States
- F32 AI102568/AI/NIAID NIH HHS/United States
- GM05551/GF/NIH HHS/United States
- T32 AI112541/AI/NIAID NIH HHS/United States
- 2T32AI112541-06/Childhood Infections Research
- T32 HL007411/HL/NHLBI NIH HHS/United States
- I01 BX005352/BX/BLRD VA/United States
LinkOut - more resources
Full Text Sources

