Glial-derived neurotrophic factor in human airway smooth muscle
- PMID: 34170009
- PMCID: PMC8671171
- DOI: 10.1002/jcp.30489
Glial-derived neurotrophic factor in human airway smooth muscle
Abstract
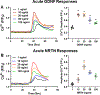
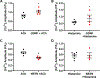
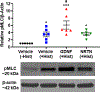
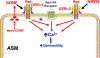
Airway smooth muscle (ASM) cells modulate the local airway milieu via production of inflammatory mediators and growth factors including classical neurotrophins, such as brain-derived neurotrophic factor (BDNF). The glial cell-derived neurotrophic factor (GDNF) family of ligands (GFLs) are nonclassical neurotrophins and their role in the airway is barely understood. The major GFLs, GDNF and Neurturin (NRTN) bind to GDNF family receptor (GFR) α1 and α2 respectively that pair with Ret receptor to accomplish signaling. In this study, we found GDNF is expressed in human lung and increased in adult asthma, while human ASM expresses GDNF and its receptors. Accordingly, we used human ASM cells to test the hypothesis that ASM expression and autocrine signaling by GFLs regulate [Ca2+ ]i . Serum-deprived ASM cells from non-asthmatics were exposed to 10 ng/ml GDNF or NRTN for 15 min (acute) or 24 h (chronic). In fura-2 loaded cells, acute GDNF or NRTN alone induced [Ca2+ ]i responses, and further enhanced responses to 1 µM ACh or 10 µM histamine. Ret inhibitor (SPP86; 10 µM) or specific GDNF chelator GFRα1-Fc (1 µg/ml) showed roles of these receptors in GDNF effects. In contrast, NRTN did not enhance [Ca2+ ]i response to histamine. Furthermore, conditioned media of nonasthmatic and asthmatic ASM cells showed GDNF secretion. SPP86, Ret inhibitor and GFRα1-Fc chelator markedly decreased [Ca2+ ]i response compared with vehicle, highlighting autocrine effects of secreted GDNF. Chronic GDNF treatment increased histamine-induced myosin light chain phosphorylation. These novel data demonstrate GFLs particularly GDNF/GFRα1 influence ASM [Ca2+ ]i and raise the possibility that GFLs are potential targets of airway hyperresponsiveness.
Keywords: Ret receptor; airway; asthma; calcium; neurotrophin.
© 2021 Wiley Periodicals LLC.
Conflict of interest statement
Figures

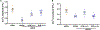
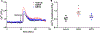
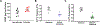
Similar articles
-
Effects of glial-derived neurotrophic factor on remodeling and mitochondrial function in human airway smooth muscle cells.Am J Physiol Lung Cell Mol Physiol. 2024 Nov 1;327(5):L684-L693. doi: 10.1152/ajplung.00101.2024. Epub 2024 Sep 24. Am J Physiol Lung Cell Mol Physiol. 2024. PMID: 39316680
-
Functional role of glial-derived neurotrophic factor in a mixed allergen murine model of asthma.Am J Physiol Lung Cell Mol Physiol. 2024 Jan 1;326(1):L19-L28. doi: 10.1152/ajplung.00099.2023. Epub 2023 Nov 21. Am J Physiol Lung Cell Mol Physiol. 2024. PMID: 37987758 Free PMC article.
-
Specificity in the crosstalk of TGFbeta/GDNF family members is determined by distinct GFR alpha receptors.J Neurochem. 2007 Dec;103(6):2491-504. doi: 10.1111/j.1471-4159.2007.04962.x. Epub 2007 Oct 22. J Neurochem. 2007. PMID: 17953664
-
Is GAS1 a co-receptor for the GDNF family of ligands?Trends Pharmacol Sci. 2006 Feb;27(2):72-7. doi: 10.1016/j.tips.2005.12.004. Epub 2006 Jan 6. Trends Pharmacol Sci. 2006. PMID: 16406089 Review.
-
Other neurotrophic factors: glial cell line-derived neurotrophic factor (GDNF).Microsc Res Tech. 1999 May 15-Jun 1;45(4-5):292-302. doi: 10.1002/(SICI)1097-0029(19990515/01)45:4/5<292::AID-JEMT13>3.0.CO;2-8. Microsc Res Tech. 1999. PMID: 10383122 Review.
Cited by
-
Evaluation of the concentration of growth associated protein-43 and glial cell-derived neurotrophic factor in degenerated intervertebral discs of the lumbosacral region of the spine.Mol Pain. 2023 Jan-Dec;19:17448069231158287. doi: 10.1177/17448069231158287. Mol Pain. 2023. PMID: 36733259 Free PMC article.
-
The central glial cell line-derived neurotrophic factor (GDNF) regulates pulmonary function in asthmatic rats.Ann Transl Med. 2023 Jan 31;11(2):113. doi: 10.21037/atm-22-6338. Ann Transl Med. 2023. PMID: 36819542 Free PMC article.
-
Downregulation of protein phosphatase 2Aα in asthmatic airway smooth muscle.Am J Physiol Lung Cell Mol Physiol. 2024 May 1;326(5):L651-L659. doi: 10.1152/ajplung.00050.2024. Epub 2024 Mar 26. Am J Physiol Lung Cell Mol Physiol. 2024. PMID: 38529552 Free PMC article.
-
Neurons enhance blood-brain barrier function via upregulating claudin-5 and VE-cadherin expression due to glial cell line-derived neurotrophic factor secretion.Elife. 2024 Oct 30;13:RP96161. doi: 10.7554/eLife.96161. Elife. 2024. PMID: 39475379 Free PMC article.
-
Effects of glial-derived neurotrophic factor on remodeling and mitochondrial function in human airway smooth muscle cells.Am J Physiol Lung Cell Mol Physiol. 2024 Nov 1;327(5):L684-L693. doi: 10.1152/ajplung.00101.2024. Epub 2024 Sep 24. Am J Physiol Lung Cell Mol Physiol. 2024. PMID: 39316680
References
-
- Amrani Y. (2007). TNF-alpha and calcium signaling in airway smooth muscle cells: a never-ending story with promising therapeutic relevance. Am J Respir Cell Mol Biol, 36(3), 387–388. Retrieved from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dop... - PubMed
-
- Appel E, Kolman O, Kazimirsky G, Blumberg PM, & Brodie C. (1997). Regulation of GDNF expression in cultured astrocytes by inflammatory stimuli. Neuroreport, 8(15), 3309–3312. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/9351662 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

