Key role of the CCR2-CCL2 axis in disease modification in a mouse model of tauopathy
- PMID: 34172073
- PMCID: PMC8234631
- DOI: 10.1186/s13024-021-00458-z
Key role of the CCR2-CCL2 axis in disease modification in a mouse model of tauopathy
Abstract
Background: For decades, dementia has been characterized by accumulation of waste in the brain and low-grade inflammation. Over the years, emerging studies highlighted the involvement of the immune system in neurodegenerative disease emergence and severity. Numerous studies in animal models of amyloidosis demonstrated the beneficial role of monocyte-derived macrophages in mitigating the disease, though less is known regarding tauopathy. Boosting the immune system in animal models of both amyloidosis and tauopathy, resulted in improved cognitive performance and in a reduction of pathological manifestations. However, a full understanding of the chain of events that is involved, starting from the activation of the immune system, and leading to disease mitigation, remained elusive. Here, we hypothesized that the brain-immune communication pathway that is needed to be activated to combat tauopathy involves monocyte mobilization via the C-C chemokine receptor 2 (CCR2)/CCL2 axis, and additional immune cells, such as CD4+ T cells, including FOXP3+ regulatory CD4+ T cells.
Methods: We used DM-hTAU transgenic mice, a mouse model of tauopathy, and applied an approach that boosts the immune system, via blocking the inhibitory Programmed cell death protein-1 (PD-1)/PD-L1 pathway, a manipulation previously shown to alleviate disease symptoms and pathology. An anti-CCR2 monoclonal antibody (αCCR2), was used to block the CCR2 axis in a protocol that partially eliminates monocytes from the circulation at the time of anti-PD-L1 antibody (αPD-L1) injection, and for the critical period of their recruitment into the brain following treatment.
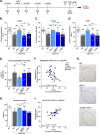
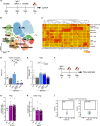
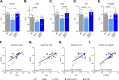
Results: Performance of DM-hTAU mice in short-term and working memory tasks, revealed that the beneficial effect of αPD-L1, assessed 1 month after a single injection, was abrogated following blockade of CCR2. This was accompanied by the loss of the beneficial effect on disease pathology, assessed by measurement of cortical aggregated human tau load using Homogeneous Time Resolved Fluorescence-based immunoassay, and by evaluation of hippocampal neuronal survival. Using both multiparametric flow cytometry, and Cytometry by Time Of Flight, we further demonstrated the accumulation of FOXP3+ regulatory CD4+ T cells in the brain, 12 days following the treatment, which was absent subsequent to CCR2 blockade. In addition, measurement of hippocampal levels of the T-cell chemoattractant, C-X-C motif chemokine ligand 12 (Cxcl12), and of inflammatory cytokines, revealed that αPD-L1 treatment reduced their expression, while blocking CCR2 reversed this effect.
Conclusions: The CCR2/CCL2 axis is required to modify pathology using PD-L1 blockade in a mouse model of tauopathy. This modification involves, in addition to monocytes, the accumulation of FOXP3+ regulatory CD4+ T cells in the brain, and the T-cell chemoattractant, Cxcl12.
Keywords: CCL2; CCR2; CXCL12; Dementia; Immunotherapy; Monocytes; PD-L1; Regulatory T cells; Tauopathy.
Conflict of interest statement
M.S. is an inventor of the intellectual property that forms the basis for development of PD-L1 immunotherapy for AD.
Figures


Similar articles
-
CCL2 Overexpression in the Brain Promotes Glial Activation and Accelerates Tau Pathology in a Mouse Model of Tauopathy.Front Immunol. 2020 May 20;11:997. doi: 10.3389/fimmu.2020.00997. eCollection 2020. Front Immunol. 2020. PMID: 32508844 Free PMC article.
-
Targeting the CCL2/CCR2 Axis in Cancer Immunotherapy: One Stone, Three Birds?Front Immunol. 2021 Nov 3;12:771210. doi: 10.3389/fimmu.2021.771210. eCollection 2021. Front Immunol. 2021. PMID: 34804061 Free PMC article. Review.
-
CCR2 Signaling Promotes Brain Infiltration of Inflammatory Monocytes and Contributes to Neuropathology during Cryptococcal Meningoencephalitis.mBio. 2021 Aug 31;12(4):e0107621. doi: 10.1128/mBio.01076-21. Epub 2021 Jul 27. mBio. 2021. PMID: 34311579 Free PMC article.
-
Blockade of CCL2/CCR2 signaling pathway prevents inflammatory monocyte recruitment and attenuates OVA-Induced allergic asthma in mice.Immunol Lett. 2019 Oct;214:30-36. doi: 10.1016/j.imlet.2019.08.006. Epub 2019 Aug 24. Immunol Lett. 2019. PMID: 31454522
-
Role of CCL2/CCR2 axis in the immunopathogenesis of rheumatoid arthritis: Latest evidence and therapeutic approaches.Life Sci. 2021 Mar 15;269:119034. doi: 10.1016/j.lfs.2021.119034. Epub 2021 Jan 13. Life Sci. 2021. PMID: 33453247 Review.
Cited by
-
Immune mechanisms and shared immune targets in neurodegenerative diseases.Nat Rev Neurol. 2025 Feb;21(2):67-85. doi: 10.1038/s41582-024-01046-7. Epub 2024 Dec 16. Nat Rev Neurol. 2025. PMID: 39681722 Review.
-
Blood and CSF chemokines in Alzheimer's disease and mild cognitive impairment: a systematic review and meta-analysis.Alzheimers Res Ther. 2023 Jun 8;15(1):107. doi: 10.1186/s13195-023-01254-1. Alzheimers Res Ther. 2023. PMID: 37291639 Free PMC article.
-
CD4+CD25+ regulatory T cell therapy in neurological autoimmune diseases.PeerJ. 2025 Jun 12;13:e19450. doi: 10.7717/peerj.19450. eCollection 2025. PeerJ. 2025. PMID: 40525111 Free PMC article. Review.
-
Dural ectopic lymphatic structures accumulate during aging and exhibit dysregulation in neurodegenerative diseases.Proc Natl Acad Sci U S A. 2025 Aug 19;122(33):e2425081122. doi: 10.1073/pnas.2425081122. Epub 2025 Aug 12. Proc Natl Acad Sci U S A. 2025. PMID: 40794835 Free PMC article.
-
Soluble PD-L1 reprograms blood monocytes to prevent cerebral edema and facilitate recovery after ischemic stroke.Brain Behav Immun. 2024 Feb;116:160-174. doi: 10.1016/j.bbi.2023.12.007. Epub 2023 Dec 7. Brain Behav Immun. 2024. PMID: 38070624 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

