Sacituzumab Govitecan for Metastatic Triple-Negative Breast Cancer: Clinical Overview and Management of Potential Toxicities
- PMID: 34176192
- PMCID: PMC8488774
- DOI: 10.1002/onco.13878
Sacituzumab Govitecan for Metastatic Triple-Negative Breast Cancer: Clinical Overview and Management of Potential Toxicities
Abstract
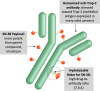
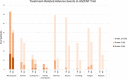
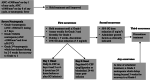
Patients with metastatic triple-negative breast cancer have a poor prognosis. Sacituzumab govitecan (IMMU-132) is an antibody-drug conjugate that contains the irinotecan active metabolite, SN-38, linked to a humanized monoclonal antibody targeting trophoblast cell surface antigen 2, which is overexpressed in many solid tumors. In a basket design phase I/II study, sacituzumab govitecan demonstrated promising single-agent therapeutic activity in multiple cancer cohorts, leading to accelerated approval by the U.S. Food and Drug Administration of sacituzumab govitecan-hziy (TRODELVY) for the treatment of patients with metastatic triple-negative breast cancer who had received at least two prior therapies in the metastatic setting. Recently, results of the phase III trial, ASCENT, were confirmatory. There is limited available information on the adverse event management with sacituzumab govitecan needed to maximize the dose and duration of effective therapy while maintaining patient quality of life. This review summarizes the clinical development and the practical management of patients receiving sacituzumab govitecan. Sacituzumab govitecan has a well-defined and manageable toxicity profile, and rapid recognition and appropriate early and proactive management will allow clinicians to optimize sacituzumab govitecan treatment for patients. IMPLICATIONS FOR PRACTICE: Sacituzumab govitecan (TRODELVY) is a novel antibody-drug conjugate composed of the active metabolite of irinotecan (SN-38) conjugated to a monoclonal antibody targeting trophoblast cell surface antigen 2, an epithelial cell surface antigen overexpressed in many cancers. Because of the rapid approval of sacituzumab govitecan, there is limited available information on adverse event (AE) management with this agent. As such, this article reviews the clinical development of the drug, the AE profile, and provides recommendations regarding AE management to help optimize therapy with sacituzumab govitecan.
Keywords: Antibody-drug conjugates; Breast cancer; Therapeutics; Toxicity.
© 2021 AlphaMed Press.
Conflict of interest statement
Disclosures of potential conflicts of interest may be found at the end of this article.
Figures

Similar articles
-
Sacituzumab Govitecan-hziy: An Antibody-Drug Conjugate for the Treatment of Refractory, Metastatic, Triple-Negative Breast Cancer.Ann Pharmacother. 2021 Jul;55(7):921-931. doi: 10.1177/1060028020966548. Epub 2020 Oct 17. Ann Pharmacother. 2021. PMID: 33070624 Review.
-
Sacituzumab govitecan: breakthrough targeted therapy for triple-negative breast cancer.Expert Rev Anticancer Ther. 2019 Aug;19(8):673-679. doi: 10.1080/14737140.2019.1654378. Epub 2019 Aug 19. Expert Rev Anticancer Ther. 2019. PMID: 31398063 Review.
-
Sacituzumab Govitecan-hziy in Refractory Metastatic Triple-Negative Breast Cancer.N Engl J Med. 2019 Feb 21;380(8):741-751. doi: 10.1056/NEJMoa1814213. N Engl J Med. 2019. PMID: 30786188 Clinical Trial.
-
Sacituzumab govitecan: past, present and future of a new antibody-drug conjugate and future horizon.Future Oncol. 2022 Sep;18(28):3199-3215. doi: 10.2217/fon-2022-0407. Epub 2022 Sep 7. Future Oncol. 2022. PMID: 36069628 Review.
-
Tackling metastatic triple-negative breast cancer with sacituzumab govitecan.Expert Rev Anticancer Ther. 2021 Dec;21(12):1303-1311. doi: 10.1080/14737140.2021.1993065. Epub 2021 Oct 22. Expert Rev Anticancer Ther. 2021. PMID: 34651524
Cited by
-
Sacituzumab Govitecan for the treatment of advanced triple negative breast cancer patients: a multi-center real-world analysis.Front Oncol. 2024 Mar 26;14:1362641. doi: 10.3389/fonc.2024.1362641. eCollection 2024. Front Oncol. 2024. PMID: 38595817 Free PMC article.
-
Preclinical evaluation of a novel antibody-drug conjugate OBI-992 for Cancer therapy.Sci Rep. 2025 Mar 13;15(1):8735. doi: 10.1038/s41598-025-92697-z. Sci Rep. 2025. PMID: 40082588 Free PMC article.
-
Escalation and optimisation of primary breast cancer treatment with antibody-drug conjugates.Transl Breast Cancer Res. 2025 Jun 18;6:23. doi: 10.21037/tbcr-25-2. eCollection 2025. Transl Breast Cancer Res. 2025. PMID: 40756955 Free PMC article. Review.
-
Targeted Therapy and Immunotherapy for Heterogeneous Breast Cancer.Cancers (Basel). 2022 Nov 6;14(21):5456. doi: 10.3390/cancers14215456. Cancers (Basel). 2022. PMID: 36358874 Free PMC article. Review.
-
Trop2-targeted therapy in breast cancer.Biomark Res. 2024 Aug 13;12(1):82. doi: 10.1186/s40364-024-00633-6. Biomark Res. 2024. PMID: 39135109 Free PMC article. Review.
References
-
- Schmid P, Adams S, Rugo HS et al. Atezolizumab and nab‐paclitaxel in advanced triple‐negative breast cancer. N Engl J Med 2018;379:2108–2121. - PubMed
-
- Puri A, Reddy TP, Patel TA et al. Metastatic triple‐negative breast cancer: Established and emerging treatments. Breast J 2020;26:1793–1796. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

