Quality of life and its predictors in adults with tuberous sclerosis complex (TSC): a multicentre cohort study from Germany
- PMID: 34176514
- PMCID: PMC8237479
- DOI: 10.1186/s42466-021-00130-3
Quality of life and its predictors in adults with tuberous sclerosis complex (TSC): a multicentre cohort study from Germany
Abstract
Background: Tuberous sclerosis complex (TSC) is a monogenetic, multisystemic disease characterised by the formation of benign tumours that can affect almost all organs, caused by pathogenic variations in TSC1 or TSC2. In this multicentre study from Germany, we investigated the influence of sociodemographic, clinical, and therapeutic factors on quality of life (QoL) among individuals with TSC.
Methods: We assessed sociodemographic and clinical characteristics and QoL among adults with TSC throughout Germany using a validated, three-month, retrospective questionnaire. We examined predictors of health-related QoL (HRQoL) using multiple linear regression analysis and compared the QoL among patients with TSC with QoL among patients with other chronic neurological disorders.
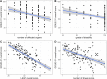
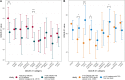
Results: We enrolled 121 adults with TSC (mean age: 31.0 ± 10.5 years; range: 18-61 years, 45.5% [n = 55] women). Unemployment, a higher grade of disability, a higher number of organ manifestations, the presence of neuropsychiatric manifestations or active epilepsy, and a higher burden of therapy-related adverse events were associated with worse QoL, as measured by two QoL instruments (EuroQoL-5 dimensions [EQ-5D] and Quality of Life in Epilepsy Patients [QOLIE-31]). Neuropsychiatric and structural nervous system manifestations, the number of affected organs, and therapy-related adverse events were also associated with higher depression, as measured by the Neurological Disorders Depression Inventory for Epilepsy (NDDI-E). In multiple regression analysis, more severe therapy-related adverse events (large effect, p < 0.001), active epilepsy (large effect, p < 0.001), and neuropsychiatric manifestations (medium effect, p = 0.003) were independently associated with worse HRQoL, explaining 65% of the variance (p < 0.001). The HRQoL among patients with active TSC-associated epilepsy was worse than that among patients with drug-refractory mesial temporal lobe epilepsy (p < 0.001), and the generic QoL among patients with more than three TSC organ manifestations was similar to those of patients with severe migraine and uncontrolled asthma.
Conclusions: Active epilepsy, neuropsychiatric manifestations (such as anxiety and depression), and therapy-related adverse events are important independent predictors of worse quality of life among adults with TSC. Generic quality of life in TSC with several manifestations is similar to uncontrolled severe chronic diseases and significantly negatively correlates with TSC severity.
Trial registration: DRKS, DRKS00016045 . Registered 01 March 2019.
Keywords: EQ-5D; Epilepsy; Organ manifestations; QOLIE-31; Quality of life; Rare disease; Seizure; TSC; Tuberous sclerosis complex; mTOR inhibitor.
Conflict of interest statement
JPZ reports speakers’ honoraria and travel grants from Eisai and Desitin Arzneimittel.
FR reports personal fees from Arvelle Therapeutics, Desitin Arzneimittel, Eisai, GW Pharmaceuticals companies, Novartis, Medtronic, Sandoz, Shire, and UCB and grants from the Detlev-Wrobel-Fonds for Epilepsy Research, the Deutsche Forschungsgemeinschaft, the LOEWE Programme of the State of Hesse, and the European Union.
MS reports personal fees from Novartis and GW Pharmaceuticals companies.
GK reports personal fees from Desitin Arzneimittel, Eisai, GW Pharmaceuticals companies, UCB, Novartis, Takeda, and Zogenix.
TM reports personal fees and grants from Arvelle Therapeutics, Eisai, GW Pharmaceuticals companies, UCB, and Zogenix.
CH reports personal fees from Desitin Arzneimittel, Eisai, GW Pharmaceuticals companies, Novartis, Shire, and Zogenix.
AB reports personal fees from Desitin Arzneimittel GmbH, Eisai GmbH, GW Pharma GmbH, Shire/Takeda GmbH, UCB Pharma GmbH, and ViroPharma GmbH.
KMK reports personal fees from UCB Pharma, Novartis Pharma AG, Eisai, and GW Pharmaceuticals and grants from the federal state Hessen, through the LOEWE program, and from the Canadian Institutes of Health Research.
SM reports grants from Novartis, UCB, Shire, Deutsche Forschungsgemeinschaft, and Epilepsiestiftung Dr. Wolf.
FvP reports personal fees and grants from Bial, Desitin Arzneimittel, Eisai, GW Pharmaceutical companies, Arvelle Therapeutics, Zogenix, and UCB Pharma.
SS-B reports personal fees from Eisai, Desitin Pharma, GW Pharmaceuticals companies, LivaNova, UCB, and Zogenix.
AS reports personal fees and grants from Arvelle Therapeutics, Desitin Arzneimittel, Eisai, GW Pharmaceutical companies, LivaNova, Marinus Pharma, Medtronic, UCB, and Zogenix.
None of the other authors reported any related conflicts of interest.
Figures

References
-
- Ebrahimi-Fakhari D, Mann LL, Poryo M, Graf N, von Kries R, Heinrich B, Ebrahimi-Fakhari D, Flotats-Bastardas M, Gortner L, Zemlin M, Meyer S. Incidence of tuberous sclerosis and age at first diagnosis: new data and emerging trends from a national, prospective surveillance study. Orphanet Journal of Rare Diseases. 2018;13(1):117. doi: 10.1186/s13023-018-0870-y. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous
