Antibiotic Resistance and Sewage-Associated Marker Genes in Untreated Sewage and a River Characterized During Baseflow and Stormflow
- PMID: 34177821
- PMCID: PMC8226142
- DOI: 10.3389/fmicb.2021.632850
Antibiotic Resistance and Sewage-Associated Marker Genes in Untreated Sewage and a River Characterized During Baseflow and Stormflow
Abstract
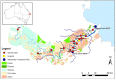
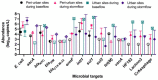
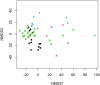
Since sewage is a hotspot for antibiotic resistance genes (ARGs), the identification of ARGs in environmental waters impacted by sewage, and their correlation to fecal indicators, is necessary to implement management strategies. In this study, sewage treatment plant (STP) influent samples were collected and analyzed using quantitative polymerase chain reaction (qPCR) to investigate the abundance and correlations between sewage-associated markers (i.e., Bacteroides HF183, Lachnospiraceae Lachno3, crAssphage) and ARGs indicating resistance to nine antibiotics (belonging to aminoglycosides, beta-lactams, sulfonamides, macrolides, and tetracyclines). All ARGs, except bla VIM, and sewage-associated marker genes were always detected in untreated sewage, and ermF and sul1 were detected in the greatest abundances. intl1 was also highly abundant in untreated sewage samples. Significant correlations were identified between sewage-associated marker genes, ARGs and the intl1 in untreated sewage (τ = 0.488, p = 0.0125). Of the three sewage-associated marker genes, the BIO-ENV procedure identified that HF183 alone best maximized correlations to ARGs and intl1 (τ = 0.590). Additionally, grab samples were collected from peri-urban and urban sites along the Brisbane River system during base and stormflow conditions, and analyzed for Escherichia coli, ARGs, the intl1, and sewage-associated marker genes using quantitative polymerase chain reaction (qPCR). Significant correlations were identified between E. coli, ARGs, and intl1 (τ = 0.0893, p = 0.0032), as well as with sewage-associated marker genes in water samples from the Brisbane River system (τ = 0.3229, p = 0.0001). Of the sewage-associated marker genes and E. coli, the BIO-ENV procedure identified that crAssphage alone maximized correlations with ARGs and intl1 in river samples (τ = 0.4148). Significant differences in E. coli, ARGs, intl1, and sewage-associated marker genes, and by flow condition (i.e., base vs. storm), and site types (peri-urban vs. urban) combined were identified (R = 0.3668, p = 0.0001), where percent dissimilarities between the multi-factorial groups ranged between 20.8 and 11.2%. Results from this study suggest increased levels of certain ARGs and sewage-associated marker genes in stormflow river water samples compared to base flow conditions. E. coli, HF183 and crAssphage may serve as potential indicators of sewage-derived ARGs under stormflow conditions, and this merits further investigation. Data presented in this study will be valuable to water quality managers to understand the links between sewage pollution and ARGs in urban environments.
Keywords: antibiotic resistance; human health risks; microbial source tracking; sewage pollution; stormwater.
Copyright © 2021 Ahmed, Gyawali, Hamilton, Joshi, Aster, Donner, Simpson and Symonds.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Source identification of antibiotic resistance genes in a peri-urban river using novel crAssphage marker genes and metagenomic signatures.Water Res. 2019 Dec 15;167:115098. doi: 10.1016/j.watres.2019.115098. Epub 2019 Sep 17. Water Res. 2019. PMID: 31574349
-
Precipitation influences pathogenic bacteria and antibiotic resistance gene abundance in storm drain outfalls in coastal sub-tropical waters.Environ Int. 2018 Jul;116:308-318. doi: 10.1016/j.envint.2018.04.005. Epub 2018 May 10. Environ Int. 2018. PMID: 29754026
-
Highly variable removal of pathogens, antibiotic resistance genes, conventional fecal indicators and human-associated fecal source markers in a pilot-scale stormwater biofilter operated under realistic stormflow conditions.Water Res. 2022 Jul 1;219:118525. doi: 10.1016/j.watres.2022.118525. Epub 2022 May 2. Water Res. 2022. PMID: 35533621
-
Fecal Pollution Drives Antibiotic Resistance and Class 1 Integron Abundance in Aquatic Environments of the Bolivian Andes Impacted by Mining and Wastewater.Microorganisms. 2020 Jul 26;8(8):1122. doi: 10.3390/microorganisms8081122. Microorganisms. 2020. PMID: 32722600 Free PMC article.
-
Occurrence of antibiotics and antibiotic resistance genes in a sewage treatment plant and its effluent-receiving river.Chemosphere. 2015 Jan;119:1379-1385. doi: 10.1016/j.chemosphere.2014.02.040. Epub 2014 Mar 12. Chemosphere. 2015. PMID: 24630248
Cited by
-
Bacterial Indicators Are Ubiquitous Members of Pelagic Microbiome in Anthropogenically Impacted Coastal Ecosystem.Front Microbiol. 2022 Jan 17;12:765091. doi: 10.3389/fmicb.2021.765091. eCollection 2021. Front Microbiol. 2022. PMID: 35111137 Free PMC article.
-
Molecular Diagnostic Tools Applied for Assessing Microbial Water Quality.Int J Environ Res Public Health. 2022 Apr 22;19(9):5128. doi: 10.3390/ijerph19095128. Int J Environ Res Public Health. 2022. PMID: 35564522 Free PMC article. Review.
-
CrAss-Like Phages: From Discovery in Human Fecal Metagenome to Application as a Microbial Source Tracking Marker.Food Environ Virol. 2024 Jun;16(2):121-135. doi: 10.1007/s12560-024-09584-5. Epub 2024 Feb 27. Food Environ Virol. 2024. PMID: 38413544 Review.
References
-
- Ahmed W., Hughes B., Harwood V. J. (2016). Current status of marker genes of Bacteroides and related taxa for identifying sewage pollution in environmental waters. Water 8:231. 10.3390/w8060231 - DOI
-
- Ahmed W., Hamilton K. A., Lobos A., Hughes B., Staley C., Sadowsky M. J., et al. (2018). Quantitative microbial risk assessment of microbial source tracking markers in recreational water contaminated with fresh untreated and secondary treated sewage. Environ. Int. 117 243–249. 10.1016/j.envint.2018.05.012 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

