Angiotensin II Receptor Blockers (ARBs Antihypertensive Agents) Increase Replication of SARS-CoV-2 in Vero E6 Cells
- PMID: 34178717
- PMCID: PMC8231006
- DOI: 10.3389/fcimb.2021.639177
Angiotensin II Receptor Blockers (ARBs Antihypertensive Agents) Increase Replication of SARS-CoV-2 in Vero E6 Cells
Abstract
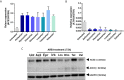
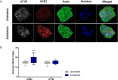
Several comorbidities, including hypertension, have been associated with an increased risk of developing severe disease during SARS-CoV-2 infection. Angiotensin II receptor blockers (ARBs) are currently some of the most widely-used drugs to control blood pressure by acting on the angiotensin II type 1 receptor (AT1R). ARBs have been reported to trigger the modulation of the angiotensin I converting enzyme 2 (ACE2), the receptor used by the virus to penetrate susceptible cells, raising concern that such treatments may promote virus capture and increase their viral load in patients receiving ARBs therapy. In this in vitro study, we reviewed the effect of ARBs on ACE2 and AT1R expression and investigated whether treatment of permissive ACE2+/AT1R+ Vero E6 cells with ARBs alters SARS-CoV-2 replication in vitro in an angiotensin II-free system. After treating the cells with the ARBs, we observed an approximate 50% relative increase in SARS-CoV-2 production in infected Vero E6 cells that correlates with the ARBs-induced up-regulation of ACE2 expression. From this data, we believe that the use of ARBs in hypertensive patients infected by SARS-CoV-2 should be carefully evaluated.
Keywords: COVID-19; SARS-CoV-2; Vero E6 cells; angiotensin I converting enzyme 2; angiotensin II receptor blockers; antihypertensive.
Copyright © 2021 Pires de Souza, Osman, Le Bideau, Baudoin, Jaafar, Devaux and La Scola.
Conflict of interest statement
CD declares owning shares in the Sanofi and Merck pharmaceutical companies. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Considerations on the use of antihypertensive blockers of the renin-angiotensin system in adults and children in the face of the COVID-19 pandemic.Bol Med Hosp Infant Mex. 2020;77(5):274-281. doi: 10.24875/BMHIM.20000158. Bol Med Hosp Infant Mex. 2020. PMID: 33064690 Review. English.
-
[ACE-inhibitors, angiotensin receptor blockers and severe acute respiratory syndrome caused by coronavirus].G Ital Cardiol (Rome). 2020 May;21(5):321-327. doi: 10.1714/3343.33127. G Ital Cardiol (Rome). 2020. PMID: 32310915 Italian.
-
RAAS Blockade and COVID-19: Mechanistic Modeling of Mas and AT1 Receptor Occupancy as Indicators of Pro-Inflammatory and Anti-Inflammatory Balance.Clin Pharmacol Ther. 2021 Apr;109(4):1092-1103. doi: 10.1002/cpt.2177. Epub 2021 Mar 10. Clin Pharmacol Ther. 2021. PMID: 33506503 Free PMC article.
-
The SARS-CoV-2 receptor, ACE-2, is expressed on many different cell types: implications for ACE-inhibitor- and angiotensin II receptor blocker-based cardiovascular therapies.Intern Emerg Med. 2020 Aug;15(5):759-766. doi: 10.1007/s11739-020-02364-6. Epub 2020 May 19. Intern Emerg Med. 2020. PMID: 32430651 Free PMC article.
-
Potential Implications of Angiotensin-converting Enzyme 2 Blockades on Neuroinflammation in SARS-CoV-2 Infection.Curr Drug Targets. 2022;23(4):364-372. doi: 10.2174/1389450122666211103165837. Curr Drug Targets. 2022. PMID: 34732115 Review.
Cited by
-
Expression of ACE2, Soluble ACE2, Angiotensin I, Angiotensin II and Angiotensin-(1-7) Is Modulated in COVID-19 Patients.Front Immunol. 2021 Jun 14;12:625732. doi: 10.3389/fimmu.2021.625732. eCollection 2021. Front Immunol. 2021. PMID: 34194422 Free PMC article.
-
Role of Angiotensin II in Cardiovascular Diseases: Introducing Bisartans as a Novel Therapy for Coronavirus 2019.Biomolecules. 2023 May 2;13(5):787. doi: 10.3390/biom13050787. Biomolecules. 2023. PMID: 37238657 Free PMC article. Review.
-
An update on angiotensin-converting enzyme 2 structure/functions, polymorphism, and duplicitous nature in the pathophysiology of coronavirus disease 2019: Implications for vascular and coagulation disease associated with severe acute respiratory syndrome coronavirus infection.Front Microbiol. 2022 Nov 28;13:1042200. doi: 10.3389/fmicb.2022.1042200. eCollection 2022. Front Microbiol. 2022. PMID: 36519165 Free PMC article. Review.
-
The Application of Mesoporous Silica Nanoparticles in Enhancing the Efficacy of Anti-Atherosclerosis Therapies: A Review.Int J Nanomedicine. 2025 Aug 10;20:9825-9856. doi: 10.2147/IJN.S538100. eCollection 2025. Int J Nanomedicine. 2025. PMID: 40821004 Free PMC article. Review.
-
Control of CDH1/E-Cadherin Gene Expression and Release of a Soluble Form of E-Cadherin in SARS-CoV-2 Infected Caco-2 Intestinal Cells: Physiopathological Consequences for the Intestinal Forms of COVID-19.Front Cell Infect Microbiol. 2022 May 4;12:798767. doi: 10.3389/fcimb.2022.798767. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35601094 Free PMC article.
References
-
- Chawla D., Rizzo S., Zalocusky K., Keebler D., Chia J., Lindsay L., et al. . (2020). Descriptive Epidemiology of 16,780 Hospitalized COVID-19 Patients in the United States. medRxiv 2020. 10.1101/2020.07.17.20156265 - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

