N-Aryl-3,4-dihydroisoquinoline Carbothioamide Analogues as Potential Urease Inhibitors
- PMID: 34179623
- PMCID: PMC8223216
- DOI: 10.1021/acsomega.1c01182
N-Aryl-3,4-dihydroisoquinoline Carbothioamide Analogues as Potential Urease Inhibitors
Abstract
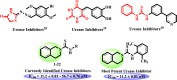
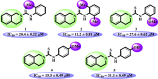
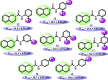
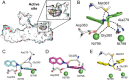
N-Aryl-3,4-dihydroisoquinoline carbothioamide analogues 1-22 were synthesized by a simple one-step reaction protocol and subjected to in vitro urease inhibition studies for the first time. All compounds 1-22 were found active and showed significant to moderate urease inhibitory potential. Specifically, analogues 1, 2, 4, and 7 were identified to be more potent (IC50 = 11.2 ± 0.81-20.4 ± 0.22 μM) than the standard thiourea (IC50 = 21.7 ± 0.34 μM). The structure-activity relationship showed that compounds bearing electron-donating groups showed superior activity. Molecular docking study on the most active derivatives revealed a good protein-ligand interaction profile against the corresponding target with key interactions, including hydrogen bonding, hydrophobic, and π-anion interactions.
© 2021 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
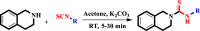
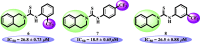
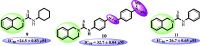
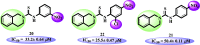
Similar articles
-
Synthetic transformation of 6-Fluoroimidazo[1,2-a]Pyridine-3-carbaldehyde into 6-Fluoroimidazo[1,2-a]Pyridine-Oxazole Derivatives: In vitro urease inhibition and in silico study.Saudi Pharm J. 2023 Aug;31(8):101667. doi: 10.1016/j.jsps.2023.05.026. Epub 2023 Jun 2. Saudi Pharm J. 2023. PMID: 37448838 Free PMC article.
-
Syntheses, in vitro urease inhibitory activities of urea and thiourea derivatives of tryptamine, their molecular docking and cytotoxic studies.Bioorg Chem. 2019 Mar;83:595-610. doi: 10.1016/j.bioorg.2018.10.070. Epub 2018 Nov 1. Bioorg Chem. 2019. PMID: 30513472
-
Synthesis, in vitro urease inhibitory activity, and molecular docking studies of thiourea and urea derivatives.Bioorg Chem. 2018 Oct;80:129-144. doi: 10.1016/j.bioorg.2018.06.007. Epub 2018 Jun 5. Bioorg Chem. 2018. PMID: 29913313
-
Synthesis of novel derivatives of oxindole, their urease inhibition and molecular docking studies.Bioorg Med Chem Lett. 2015 Aug 15;25(16):3285-9. doi: 10.1016/j.bmcl.2015.05.069. Epub 2015 May 30. Bioorg Med Chem Lett. 2015. PMID: 26077497
-
2-(Hetero(aryl)methylene)hydrazine-1-carbothioamides as potent urease inhibitors.Chem Biol Drug Des. 2015 Feb;85(2):225-30. doi: 10.1111/cbdd.12379. Epub 2014 Jul 10. Chem Biol Drug Des. 2015. PMID: 24938644
Cited by
-
Design and synthesis of new N-thioacylated ciprofloxacin derivatives as urease inhibitors with potential antibacterial activity.Sci Rep. 2022 Aug 15;12(1):13827. doi: 10.1038/s41598-022-17993-4. Sci Rep. 2022. PMID: 35970866 Free PMC article.
-
Thioamides in medicinal chemistry and as small molecule therapeutic agents.Eur J Med Chem. 2024 Nov 5;277:116732. doi: 10.1016/j.ejmech.2024.116732. Epub 2024 Aug 5. Eur J Med Chem. 2024. PMID: 39106658 Free PMC article. Review.
-
Synthesis, Biological Evaluation, and Molecular Dynamics of Carbothioamides Derivatives as Carbonic Anhydrase II and 15-Lipoxygenase Inhibitors.Molecules. 2022 Dec 9;27(24):8723. doi: 10.3390/molecules27248723. Molecules. 2022. PMID: 36557863 Free PMC article.
-
Synthesis, Urease Inhibition, Molecular Docking, and Optical Analysis of a Symmetrical Schiff Base and Its Selected Metal Complexes.Molecules. 2024 Oct 16;29(20):4899. doi: 10.3390/molecules29204899. Molecules. 2024. PMID: 39459267 Free PMC article.
References
-
- Pervez H.; Iqbal M. S.; Tahir M. Y.; Nasim F. U. H.; Choudhary M. I.; Khan K. M. In vitro cytotoxic, antibacterial, antifungal and urease inhibitory activities of some N4-substituted isatin-3-thiosemicarbazones. J. Enzymem Inhib. Med. Chem. 2008, 23, 848–854. 10.1080/14756360701746179. - DOI - PubMed
-
- Arshad T.; Khan K. M.; Rasool N.; Salar U.; Hussain S.; Asghar H.; Ashraf M.; Wadood A.; Riaz M.; Perveen S.; Taha M.; Ismail N. H. 5-Bromo-2-aryl benzimidazole derivatives as non-cytotoxic potential dual inhibitors of α-glucosidase and urease enzymes. Bioorg. Chem. 2017, 72, 21–31. 10.1016/j.bioorg.2017.03.007. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

