Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV): Designing Master Protocols for Evaluation of Candidate COVID-19 Therapeutics
- PMID: 34181444
- PMCID: PMC8244665
- DOI: 10.7326/M21-1269
Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV): Designing Master Protocols for Evaluation of Candidate COVID-19 Therapeutics
Abstract
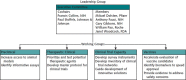
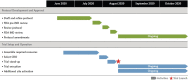
Working in an unprecedented time frame, the Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV) public-private partnership developed and launched 9 master protocols between 14 April 2020 and 31 May 2021 to allow for the coordinated and efficient evaluation of multiple investigational therapeutic agents for COVID-19. The ACTIV master protocols were designed with a portfolio approach to serve the following patient populations with COVID-19: mild to moderately ill outpatients, moderately ill inpatients, and critically ill inpatients. To facilitate the execution of these studies and minimize start-up time, ACTIV selected several existing networks to launch the master protocols. The master protocols were also designed to test several agent classes prioritized by ACTIV that covered the spectrum of the disease pathophysiology. Each protocol, either adaptive or pragmatic, was designed to efficiently select those treatments that provide benefit to patients while rapidly eliminating those that were either ineffective or unsafe. The ACTIV Therapeutics-Clinical Working Group members describe the process by which these master protocols were designed, developed, and launched. Lessons learned that may be useful in meeting the challenges of a future pandemic are also described.
Conflict of interest statement
Figures






References
-
- Nanda R , Liu MC , Yau C , et al. Effect of pembrolizumab plus neoadjuvant chemotherapy on pathologic complete response in women with early-stage breast cancer: an analysis of the ongoing phase 2 adaptively randomized I-SPY2 trial. JAMA Oncol. 2020;6:676-684. [PMID: ] doi: 10.1001/jamaoncol.2019.6650 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
