TM4SF5 Knockout Protects Mice From Diet-Induced Obesity Partly by Regulating Autophagy in Adipose Tissue
- PMID: 34187836
- PMCID: PMC8576418
- DOI: 10.2337/db21-0145
TM4SF5 Knockout Protects Mice From Diet-Induced Obesity Partly by Regulating Autophagy in Adipose Tissue
Abstract
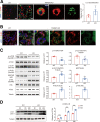
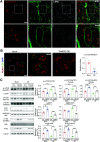
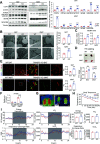
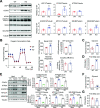
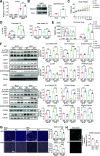
Transmembrane 4 L six family member 5 (TM4SF5) functions as a sensor for lysosomal arginine levels and activates the mammalian target of rapamycin complex 1 (mTORC1). While the mTORC1 signaling pathway plays a key role in adipose tissue metabolism, the regulatory function of TM4SF5 in adipocytes remains unclear. In this study we aimed to establish a TM4SF5 knockout (KO) mouse model and investigated the effects of TM4SF5 KO on mTORC1 signaling-mediated autophagy and mitochondrial metabolism in adipose tissue. TM4SF5 expression was higher in inguinal white adipose tissue (iWAT) than in brown adipose tissue and significantly upregulated by a high-fat diet (HFD). TM4SF5 KO reduced mTORC1 activation and enhanced autophagy and lipolysis in adipocytes. RNA sequencing analysis of TM4SF5 KO mouse iWAT showed that the expression of genes involved in peroxisome proliferator-activated receptor α signaling pathways and mitochondrial oxidative metabolism was upregulated. Consequently, TM4SF5 KO reduced adiposity and increased energy expenditure and mitochondrial oxidative metabolism. TM4SF5 KO prevented HFD-induced glucose intolerance and inflammation in adipose tissue. Collectively, the results of our study demonstrate that TM4SF5 regulates autophagy and lipid catabolism in adipose tissue and suggest that TM4SF5 could be therapeutically targeted for the treatment of obesity-related metabolic diseases.
© 2021 by the American Diabetes Association.
Figures
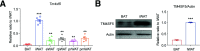


Similar articles
-
Sustained activation of autophagy suppresses adipocyte maturation via a lipolysis-dependent mechanism.Autophagy. 2020 Sep;16(9):1668-1682. doi: 10.1080/15548627.2019.1703355. Epub 2019 Dec 18. Autophagy. 2020. PMID: 31840569 Free PMC article.
-
GIP receptor deletion in mice confers resistance to high-fat diet-induced obesity via alterations in energy expenditure and adipose tissue lipid metabolism.Am J Physiol Endocrinol Metab. 2021 Apr 1;320(4):E835-E845. doi: 10.1152/ajpendo.00646.2020. Epub 2021 Mar 1. Am J Physiol Endocrinol Metab. 2021. PMID: 33645252
-
REEP6 knockout leads to defective β-adrenergic signaling in adipocytes and promotes obesity-related metabolic dysfunction.Metabolism. 2022 May;130:155159. doi: 10.1016/j.metabol.2022.155159. Epub 2022 Feb 9. Metabolism. 2022. PMID: 35150731
-
mTORC1 in energy expenditure: consequences for obesity.Nat Rev Endocrinol. 2024 Apr;20(4):239-251. doi: 10.1038/s41574-023-00934-0. Epub 2024 Jan 15. Nat Rev Endocrinol. 2024. PMID: 38225400 Review.
-
Amino acid transporters as tetraspanin TM4SF5 binding partners.Exp Mol Med. 2020 Jan;52(1):7-14. doi: 10.1038/s12276-019-0363-7. Epub 2020 Jan 20. Exp Mol Med. 2020. PMID: 31956272 Free PMC article. Review.
Cited by
-
High-fiber-diet-related metabolites improve neurodegenerative symptoms in patients with obesity with diabetes mellitus by modulating the hippocampal-hypothalamic endocrine axis.Front Neurol. 2023 Jan 17;13:1026904. doi: 10.3389/fneur.2022.1026904. eCollection 2022. Front Neurol. 2023. PMID: 36733447 Free PMC article.
-
TM4SF5-mediated abnormal food-intake behavior and apelin expression facilitate non-alcoholic fatty liver disease features.iScience. 2023 Aug 14;26(9):107625. doi: 10.1016/j.isci.2023.107625. eCollection 2023 Sep 15. iScience. 2023. PMID: 37670786 Free PMC article.
-
BNIP3-mediated mitophagy in macrophages regulates obesity-induced adipose tissue metaflammation.Autophagy. 2025 Sep;21(9):2009-2027. doi: 10.1080/15548627.2025.2487035. Epub 2025 Apr 22. Autophagy. 2025. PMID: 40195021 Free PMC article.
-
PathGPS: discover shared genetic architecture using GWAS summary data.Biometrics. 2024 Jul 1;80(3):ujae060. doi: 10.1093/biomtc/ujae060. Biometrics. 2024. PMID: 39005072 Free PMC article.
-
Liver-originated small extracellular vesicles with TM4SF5 target brown adipose tissue for homeostatic glucose clearance.J Extracell Vesicles. 2022 Sep;11(9):e12262. doi: 10.1002/jev2.12262. J Extracell Vesicles. 2022. PMID: 36063136 Free PMC article.
References
-
- Saely CH, Geiger K, Drexel H. Brown versus white adipose tissue: a mini-review. Gerontology 2012;58:15–23 - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

