The bacterial toxin ExoU requires a host trafficking chaperone for transportation and to induce necrosis
- PMID: 34188051
- PMCID: PMC8241856
- DOI: 10.1038/s41467-021-24337-9
The bacterial toxin ExoU requires a host trafficking chaperone for transportation and to induce necrosis
Abstract
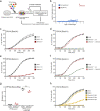
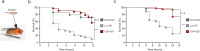
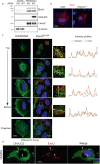
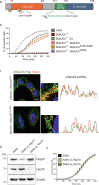
Pseudomonas aeruginosa can cause nosocomial infections, especially in ventilated or cystic fibrosis patients. Highly pathogenic isolates express the phospholipase ExoU, an effector of the type III secretion system that acts on plasma membrane lipids, causing membrane rupture and host cell necrosis. Here, we use a genome-wide screen to discover that ExoU requires DNAJC5, a host chaperone, for its necrotic activity. DNAJC5 is known to participate in an unconventional secretory pathway for misfolded proteins involving anterograde vesicular trafficking. We show that DNAJC5-deficient human cells, or Drosophila flies knocked-down for the DNAJC5 orthologue, are largely resistant to ExoU-dependent virulence. ExoU colocalizes with DNAJC5-positive vesicles in the host cytoplasm. DNAJC5 mutations preventing vesicle trafficking (previously identified in adult neuronal ceroid lipofuscinosis, a human congenital disease) inhibit ExoU-dependent cell lysis. Our results suggest that, once injected into the host cytoplasm, ExoU docks to DNAJC5-positive secretory vesicles to reach the plasma membrane, where it can exert its phospholipase activity.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Abnormal triaging of misfolded proteins by adult neuronal ceroid lipofuscinosis-associated DNAJC5/CSPα mutants causes lipofuscin accumulation.Autophagy. 2023 Jan;19(1):204-223. doi: 10.1080/15548627.2022.2065618. Epub 2022 May 4. Autophagy. 2023. PMID: 35506243 Free PMC article.
-
Intoxication of host cells by the T3SS phospholipase ExoU: PI(4,5)P2-associated, cytoskeletal collapse and late phase membrane blebbing.PLoS One. 2014 Jul 25;9(7):e103127. doi: 10.1371/journal.pone.0103127. eCollection 2014. PLoS One. 2014. PMID: 25061861 Free PMC article.
-
Structural basis of cytotoxicity mediated by the type III secretion toxin ExoU from Pseudomonas aeruginosa.PLoS Pathog. 2012;8(4):e1002637. doi: 10.1371/journal.ppat.1002637. Epub 2012 Apr 5. PLoS Pathog. 2012. PMID: 22496657 Free PMC article.
-
Perspectives on the Pseudomonas aeruginosa Type III Secretion System Effector ExoU and Its Subversion of the Host Innate Immune Response to Infection.Toxins (Basel). 2021 Dec 9;13(12):880. doi: 10.3390/toxins13120880. Toxins (Basel). 2021. PMID: 34941717 Free PMC article. Review.
-
Association between Pseudomonas aeruginosa type III secretion, antibiotic resistance, and clinical outcome: a review.Crit Care. 2014 Dec 13;18(6):668. doi: 10.1186/s13054-014-0668-9. Crit Care. 2014. PMID: 25672496 Free PMC article. Review.
Cited by
-
N-terminal signal peptides facilitate the engineering of PVC complex as a potent protein delivery system.Sci Adv. 2022 Apr 29;8(17):eabm2343. doi: 10.1126/sciadv.abm2343. Epub 2022 Apr 29. Sci Adv. 2022. PMID: 35486720 Free PMC article.
-
Lipase-mediated detoxification of host-derived antimicrobial fatty acids by Staphylococcus aureus.Commun Biol. 2024 May 15;7(1):572. doi: 10.1038/s42003-024-06278-3. Commun Biol. 2024. PMID: 38750133 Free PMC article.
-
Host phospholipid peroxidation fuels ExoU-dependent cell necrosis and supports Pseudomonas aeruginosa-driven pathology.PLoS Pathog. 2021 Sep 13;17(9):e1009927. doi: 10.1371/journal.ppat.1009927. eCollection 2021 Sep. PLoS Pathog. 2021. PMID: 34516571 Free PMC article.
-
RetS-mediated environmental sensing coordinates TetR-dependent regulation of type III secretion system and virulence in Pseudomonas syringae pv. actinidiae.Appl Environ Microbiol. 2025 Jul 23;91(7):e0049425. doi: 10.1128/aem.00494-25. Epub 2025 Jun 10. Appl Environ Microbiol. 2025. PMID: 40492733 Free PMC article.
-
Cell lipid biology in infections: an overview.Front Cell Infect Microbiol. 2023 Oct 6;13:1148383. doi: 10.3389/fcimb.2023.1148383. eCollection 2023. Front Cell Infect Microbiol. 2023. PMID: 37868347 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

