Passive transfer of fibromyalgia symptoms from patients to mice
- PMID: 34196305
- PMCID: PMC8245181
- DOI: 10.1172/JCI144201
Passive transfer of fibromyalgia symptoms from patients to mice
Abstract
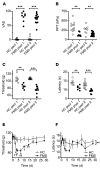
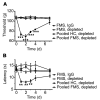
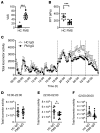
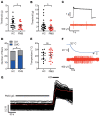
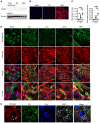
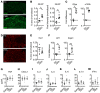
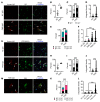
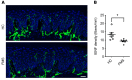
Fibromyalgia syndrome (FMS) is characterized by widespread pain and tenderness, and patients typically experience fatigue and emotional distress. The etiology and pathophysiology of fibromyalgia are not fully explained and there are no effective drug treatments. Here we show that IgG from FMS patients produced sensory hypersensitivity by sensitizing nociceptive neurons. Mice treated with IgG from FMS patients displayed increased sensitivity to noxious mechanical and cold stimulation, and nociceptive fibers in skin-nerve preparations from mice treated with FMS IgG displayed an increased responsiveness to cold and mechanical stimulation. These mice also displayed reduced locomotor activity, reduced paw grip strength, and a loss of intraepidermal innervation. In contrast, transfer of IgG-depleted serum from FMS patients or IgG from healthy control subjects had no effect. Patient IgG did not activate naive sensory neurons directly. IgG from FMS patients labeled satellite glial cells and neurons in vivo and in vitro, as well as myelinated fiber tracts and a small number of macrophages and endothelial cells in mouse dorsal root ganglia (DRG), but no cells in the spinal cord. Furthermore, FMS IgG bound to human DRG. Our results demonstrate that IgG from FMS patients produces painful sensory hypersensitivities by sensitizing peripheral nociceptive afferents and suggest that therapies reducing patient IgG titers may be effective for fibromyalgia.
Keywords: Autoimmune diseases; Neurological disorders; Neuroscience; Pain.
Conflict of interest statement
Figures



Comment in
-
From human to mouse and back offers hope for patients with fibromyalgia.J Clin Invest. 2021 Jul 1;131(13):e150382. doi: 10.1172/JCI150382. J Clin Invest. 2021. PMID: 34196306 Free PMC article.
-
Antibodies induce fibromyalgia symptoms.Nat Rev Rheumatol. 2021 Sep;17(9):507. doi: 10.1038/s41584-021-00679-y. Nat Rev Rheumatol. 2021. PMID: 34354268 No abstract available.
References
-
- Kosek E, et al. Sensory dysfunction in fibromyalgia patients with implications for pathogenic mechanisms. Pain. 1996;68(2–3):375–383. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

