Multi-Target Directed Ligands (MTDLs) Binding the σ1 Receptor as Promising Therapeutics: State of the Art and Perspectives
- PMID: 34198620
- PMCID: PMC8232171
- DOI: 10.3390/ijms22126359
Multi-Target Directed Ligands (MTDLs) Binding the σ1 Receptor as Promising Therapeutics: State of the Art and Perspectives
Abstract
The sigma-1 (σ1) receptor is a 'pluripotent chaperone' protein mainly expressed at the mitochondria-endoplasmic reticulum membrane interfaces where it interacts with several client proteins. This feature renders the σ1 receptor an ideal target for the development of multifunctional ligands, whose benefits are now recognized because several pathologies are multifactorial. Indeed, the current therapeutic regimens are based on the administration of different classes of drugs in order to counteract the diverse unbalanced physiological pathways associated with the pathology. Thus, the multi-targeted directed ligand (MTDL) approach, with one molecule that exerts poly-pharmacological actions, may be a winning strategy that overcomes the pharmacokinetic issues linked to the administration of diverse drugs. This review aims to point out the progress in the development of MTDLs directed toward σ1 receptors for the treatment of central nervous system (CNS) and cancer diseases, with a focus on the perspectives that are proper for this strategy. The evidence that some drugs in clinical use unintentionally bind the σ1 protein (as off-target) provides a proof of concept of the potential of this strategy, and it strongly supports the promise that the σ1 receptor holds as a target to be hit in the context of MTDLs for the therapy of multifactorial pathologies.
Keywords: multi-target directed ligands (MTDLs); polypharmacology; sigma receptors; sigma-1 ligands; sigma-1 receptor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

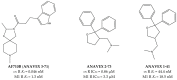


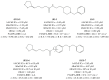


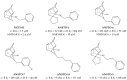

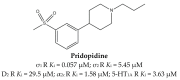

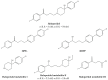
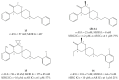
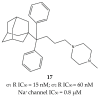


References
-
- Walker J.M., Bowen W.D., Walker F.O., Matsumoto R.R., De Costa B., Rice K.C. Sigma Receptors: Biology and Function. Pharmacol. Rev. 1990;42:355–402. - PubMed
-
- Hellewell S.B., Bowen W.D. A Sigma-like Binding Site in Rat Pheochromocytoma (PC12) Cells: Decreased Affinity for (+)-Benzomorphans and Lower Molecular Weight Suggest a Different Sigma Receptor Form from That of Guinea Pig Brain. Brain Res. 1990;527:244–253. doi: 10.1016/0006-8993(90)91143-5. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

