Emerging Immunogenicity and Genotoxicity Considerations of Adeno-Associated Virus Vector Gene Therapy for Hemophilia
- PMID: 34199563
- PMCID: PMC8199697
- DOI: 10.3390/jcm10112471
Emerging Immunogenicity and Genotoxicity Considerations of Adeno-Associated Virus Vector Gene Therapy for Hemophilia
Abstract
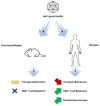
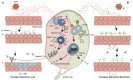
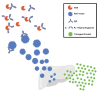
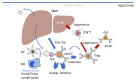
Adeno-associated viral (AAV) vector gene therapy has shown promise as a possible cure for hemophilia. However, immune responses directed against AAV vectors remain a hurdle to the broader use of this gene transfer platform. Both innate and adaptive immune responses can affect the safety and efficacy of AAV vector-mediated gene transfer in humans. These immune responses may be triggered by the viral capsid, the vector's nucleic acid payload, or other vector contaminants or excipients, or by the transgene product encoded by the vector itself. Various preclinical and clinical strategies have been explored to overcome the issues of AAV vector immunogenicity and transgene-related immune responses. Although results of these strategies are encouraging, more efficient approaches are needed to deliver safe, predictable, and durable outcomes for people with hemophilia. In addition to durability, long-term follow-up of gene therapy trial participants will allow us to address potential safety concerns related to vector integration. Herein, we describe the challenges with current methodologies to deliver optimal outcomes for people with hemophilia who choose to undergo AAV vector gene therapy and the potential opportunities to improve on the results.
Keywords: adeno-associated virus vector; cellular immunity; gene therapy; genome integration; hemophilia; humoral immunity; immunogenicity; immunologic tolerance; innate immunity; liver transduction; neutralizing antibody.
Conflict of interest statement
P.E.M. reports that during a portion of the preparation of the manuscript, P.E.M. was an employee of Spark Therapeutics. C.N. reports receipt of grants/research supports or honoraria/consultation fees from Bayer, CSL Behring, Freeline, LFB, Novo Nordisk, Octapharma, Pfizer, Roche, Sanofi, Sobi™, Spark Therapeutics, Takeda. M.T. reports consultancy fees from BioMarin, Genentech, Pfizer, Octapharma, Spark Therapeutics, and Takeda; Speaker’s bureau fees for BioMarin, Genentech, Octapharma, Grifols, and Takeda; clinical trial principal investigator for Pfizer, Spark Therapeutics, and Takeda; grant review fees from Pfizer; receipt of research grant from Grifols; salaried position at the Bleeding and Clotting Disorders Institute; private practice Michael D. Tarantino, MD, SC. L.A.V. reports was an employee and stock holder of Spark Therapeutics prior to the development of this manuscript. F.M. reports is an employee of Spark Therapeutics, outside the submitted work.
Figures


References
-
- Konkle B.A., Walsh C.E., Escobar M.A., Josephson N.C., Young G., von Drygalski A., McPhee S.W.J., Samulski R.J., Bilic I., de la Rosa M., et al. BAX 335 hemophilia B gene therapy clinical trial results: Potential impact of CpG sequences on gene expression. Blood. 2021;137:763–774. doi: 10.1182/blood.2019004625. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

