Feasibility, Safety and Impact on Overall Survival of Awake Resection for Newly Diagnosed Supratentorial IDH-Wildtype Glioblastomas in Adults
- PMID: 34200799
- PMCID: PMC8230499
- DOI: 10.3390/cancers13122911
Feasibility, Safety and Impact on Overall Survival of Awake Resection for Newly Diagnosed Supratentorial IDH-Wildtype Glioblastomas in Adults
Abstract
Background: Although awake resection using intraoperative cortico-subcortical functional brain mapping is the benchmark technique for diffuse gliomas within eloquent brain areas, it is still rarely proposed for IDH-wildtype glioblastomas. We have assessed the feasibility, safety, and efficacy of awake resection for IDH-wildtype glioblastomas.
Methods: Observational single-institution cohort (2012-2018) of 453 adult patients harboring supratentorial IDH-wildtype glioblastomas who benefited from awake resection, from asleep resection, or from a biopsy. Case matching (1:1) criteria between the awake group and asleep group: gender, age, RTOG-RPA class, tumor side, location and volume and neurosurgeon experience.
Results: In patients in the awake resection subgroup (n = 42), supratotal resections were more frequent (21.4% vs. 3.1%, p < 0.0001) while partial resections were less frequent (21.4% vs. 40.1%, p < 0.0001) compared to the asleep (n = 222) resection subgroup. In multivariable analyses, postoperative standard radiochemistry (aHR = 0.04, p < 0.0001), supratotal resection (aHR = 0.27, p = 0.0021), total resection (aHR = 0.43, p < 0.0001), KPS score > 70 (HR = 0.66, p = 0.0013), MGMT promoter methylation (HR = 0.55, p = 0.0031), and awake surgery (HR = 0.54, p = 0.0156) were independent predictors of overall survival. After case matching, a longer overall survival was found for awake resection (HR = 0.47, p = 0.0103).
Conclusions: Awake resection is safe, allows larger resections than asleep surgery, and positively impacts overall survival of IDH-wildtype glioblastoma in selected adult patients.
Keywords: IDH-wildtype; awake surgery; extent of resection; glioblastoma; survival.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
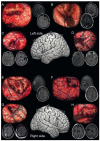
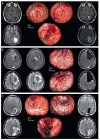

References
-
- Molinaro A.M., Hervey-Jumper S., Morshed R.A., Young J., Han S.J., Chunduru P., Zhang Y., Phillips J.J., Shai A., Lafontaine M., et al. Association of maximal extent of resection of contrast-enhanced and non–contrast-enhanced tumor with survival within molecular subgroups of patients with newly diagnosed glioblastoma. JAMA Oncol. 2020;6:495–503. doi: 10.1001/jamaoncol.2019.6143. - DOI - PMC - PubMed
-
- Chambless L.B., Kistka H.M., Parker S.L., Hassam-Malani L., McGirt M.J., Thompson R.C., Mohammad L. The relative value of postoperative versus preoperative Karnofsky Performance Scale scores as a predictor of survival after surgical resection of glioblastoma multiforme. J. Neuro Oncol. 2014;121:359–364. doi: 10.1007/s11060-014-1640-x. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

