Antibacterial Activity of a Promising Antibacterial Agent: 22-(4-(2-(4-Nitrophenyl-piperazin-1-yl)-acetyl)-piperazin-1-yl)-22-deoxypleuromutilin
- PMID: 34201372
- PMCID: PMC8227856
- DOI: 10.3390/molecules26123502
Antibacterial Activity of a Promising Antibacterial Agent: 22-(4-(2-(4-Nitrophenyl-piperazin-1-yl)-acetyl)-piperazin-1-yl)-22-deoxypleuromutilin
Abstract
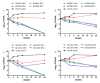
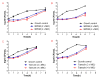
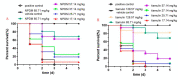
A novel pleuromutilin derivative, 22-(4-(2-(4-nitrophenyl-piperazin-1-yl)-acetyl)-piperazin-1-yl)-22-deoxypleuromutilin (NPDM), was synthesized in our laboratory and proved excellent antibacterial activity against methicillin-resistant Staphylococcus aureus (MRSA). In this study, more methods were used to further study its preliminary pharmacological effect. The antibacterial efficacy and toxicity of NPDM were evaluated using tiamulin as the reference drug. The in vitro antibacterial activity study showed that NPDM is a potent bactericidal agent against MRSA that induced time-dependent growth inhibition and a concentration-dependent post-antibiotic effect (PAE). Toxicity determination showed that the cytotoxicity of NPDM was slightly higher than that of tiamulin, but the acute oral toxicity study proved that NPDM was a low-toxic compound. In an in vivo antibacterial effect study, NPDM exhibited a better therapeutic effect than tiamulin against MRSA in a mouse thigh infection model as well as a mouse systemic infection model with neutropenia. The 50% effective dose (ED50) of NPDM in a Galleria mellonella infection model was 50.53 mg/kg. The pharmacokinetic properties of NPDM were also measured, which showed that NPDM was a rapid elimination drug in mice.
Keywords: LC-MS/MS; MRSA; antibacterial activity; pharmacokinetic; pleuromutilin derivative.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Kaku N., Yanagihara K., Morinaga Y., Yamada K., Harada Y., Migiyama Y., Nagaoka K., Matsuda J.-I., Uno N., Hasegawa H., et al. Influence of antimicrobial regimen on decreased in-hospital mortality of patients with MRSA bacteremia. J. Infect. Chemother. 2014;20:350–355. doi: 10.1016/j.jiac.2013.12.009. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

