Curcuma longa L. Water Extract Improves Dexamethasone-Induced Sarcopenia by Modulating the Muscle-Related Gene and Oxidative Stress in Mice
- PMID: 34201533
- PMCID: PMC8300838
- DOI: 10.3390/antiox10071000
Curcuma longa L. Water Extract Improves Dexamethasone-Induced Sarcopenia by Modulating the Muscle-Related Gene and Oxidative Stress in Mice
Abstract
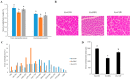
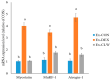
Dexamethasone (DEX) promotes proteolysis, which causes muscle atrophy. Muscle atrophy is connected to sarcopenia. We evaluated the effect of Curcuma longa L. water extract (CLW) on DEX-induced muscle atrophy. ICR mice were divided into three groups (eight mice per group) to investigate the capability of CLW in inhibiting muscle atrophy. The control group (Ex-CON) was administered distilled water (DW) by gavage and subjected to exercise; the muscle atrophy group (Ex-DEX) was administered DW by gavage, an injection of DEX (1 mg/kg body weight/day) intraperitoneally (IP), and subjected to exercise; and the treatment group (Ex-CLW) was administered CLW (1 g/kg body weight/day) by gavage, DEX IP injection, and subjected to exercise. Following the injection of DEX, the expression levels of myostatin, MuRF-1, and Atrogin-1 were increased. However, these expression levels were decreased in the Ex-CLW group, thereby leading to the conclusion that CLW inhibits muscle atrophy. ROS (that was overproduced by DEX) decreased antioxidant enzyme activity and increased malondialdehyde (MDA) levels, which led to muscle atrophy. When CLW was ingested, the antioxidant enzyme activities increased while the MDA levels decreased. These findings suggest that CLW could serve as a natural product for the prevention of muscle atrophy by modulating muscle atrophy-related genes and increasing antioxidant potential.
Keywords: Curcuma longa L.; antioxidants; ladder climbing exercise; muscle function; sarcopenia.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Yamamoto D., Maki T., Herningtyas E.H., Ikeshita N., Shibahara H., Sugiyzma Y., Nakanishi S., Iida K., Iguchi G., Takahashi Y., et al. Branched-Chain amino acids protect against dexamethasone-induced soleus muscle atrophy in rats. Muscle Nerve. 2010;41:819–827. doi: 10.1002/mus.21621. - DOI - PubMed
LinkOut - more resources
Full Text Sources

