Alnus sibirica Compounds Exhibiting Anti-Proliferative, Apoptosis-Inducing, and GSTP1 Demethylating Effects on Prostate Cancer Cells
- PMID: 34201712
- PMCID: PMC8270241
- DOI: 10.3390/molecules26133830
Alnus sibirica Compounds Exhibiting Anti-Proliferative, Apoptosis-Inducing, and GSTP1 Demethylating Effects on Prostate Cancer Cells
Abstract
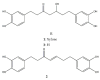
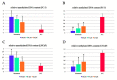
Alnus sibirica (AS) is distributed in Korea, Japan, China, and Russia and has reported anti-oxidant, anti-inflammatory, and reducing activities on atopic dermatitis-like skin lesions, along with other beneficial health properties. In the present study, we tried to prove the cancer-preventive activity against prostate cancer. The extracted and isolated compounds, oregonin (1), hirsutenone (2), and hirsutanonol (3), which were isolated from AS, were tested for anti-proliferative activity. To do this, we used the MTT assay; NF-κB inhibitory activity, using Western blotting; apoptosis-inducing activity using flow cytometry; DNA methylation activity, using methylation-specific polymerase chain reaction in androgen-dependent (LNCaP) and androgen-independent (PC-3) prostate cancer cell lines. The compounds (1-3) showed potent anti-proliferative activity against both prostate cancer cell lines. Hirsutenone (2) exhibited the strongest NF-κB inhibitory and apoptosis-inducing activities compared with oregonin (1) and hirsutanonol (3). DNA methylation activity, which was assessed for hirsutenone (2), revealed a concentration-dependent enhancement of the unmethylated DNA content and a reduction in the methylated DNA content in both PC-3 and LNCaP cells. Overall, these findings suggest that hirsutenone (2), when isolated from AS, may be a potential agent for preventing the development or progression of prostate cancer.
Keywords: Alnus sibirica; DNA methylation; cancer prevention; hirsutenone; prostate cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Hirsutenone in Alnus extract inhibits akt activity and suppresses prostate cancer cell proliferation.Mol Carcinog. 2015 Nov;54(11):1354-62. doi: 10.1002/mc.22211. Epub 2014 Sep 11. Mol Carcinog. 2015. PMID: 25213146
-
The diarylheptanoid hirsutenone sensitizes chemoresistant ovarian cancer cells to cisplatin via modulation of apoptosis-inducing factor and X-linked inhibitor of apoptosis.J Biol Chem. 2014 Jan 17;289(3):1723-31. doi: 10.1074/jbc.M113.513879. Epub 2013 Nov 18. J Biol Chem. 2014. PMID: 24247248 Free PMC article.
-
Inhibitory Effects on NO Production and DPPH Radicals and NBT Superoxide Activities of Diarylheptanoid Isolated from Enzymatically Hydrolyzed Ehthanolic Extract of Alnus sibirica.Molecules. 2019 May 20;24(10):1938. doi: 10.3390/molecules24101938. Molecules. 2019. PMID: 31137531 Free PMC article.
-
Antiproliferative, apoptosis-inducing, and GSTP1 demethylation activities of Ellagitannins isolated from Cornus alba L.Nat Prod Res. 2025 Mar;39(5):1411-1417. doi: 10.1080/14786419.2023.2295918. Epub 2023 Dec 22. Nat Prod Res. 2025. PMID: 38135905
-
Diarylheptanoid hirsutenone prevents tumor necrosis factor-alpha-stimulated production of inflammatory mediators in human keratinocytes through NF-kappaB inhibition.Int Immunopharmacol. 2009 Aug;9(9):1097-104. doi: 10.1016/j.intimp.2009.05.006. Epub 2009 May 20. Int Immunopharmacol. 2009. PMID: 19464389
Cited by
-
Can Natural Products Targeting EMT Serve as the Future Anticancer Therapeutics?Molecules. 2022 Nov 8;27(22):7668. doi: 10.3390/molecules27227668. Molecules. 2022. PMID: 36431766 Free PMC article. Review.
-
Resistomycin Suppresses Prostate Cancer Cell Growth by Instigating Oxidative Stress, Mitochondrial Apoptosis, and Cell Cycle Arrest.Molecules. 2023 Nov 30;28(23):7871. doi: 10.3390/molecules28237871. Molecules. 2023. PMID: 38067602 Free PMC article.
-
Biological Activity of Natural and Synthetic Compounds.Molecules. 2022 Jun 7;27(12):3652. doi: 10.3390/molecules27123652. Molecules. 2022. PMID: 35744778 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

