Nucleocytoplasmic Trafficking Perturbation Induced by Picornaviruses
- PMID: 34201715
- PMCID: PMC8310216
- DOI: 10.3390/v13071210
Nucleocytoplasmic Trafficking Perturbation Induced by Picornaviruses
Abstract
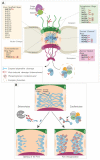
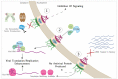
Picornaviruses are positive-stranded RNA viruses. Even though replication and translation of their genome take place in the cytoplasm, these viruses evolved different strategies to disturb nucleocytoplasmic trafficking of host proteins and RNA. The major targets of picornavirus are the phenylalanine-glycine (FG)-nucleoporins, which form a mesh in the central channel of the nuclear pore complex through which protein cargos and karyopherins are actively transported in both directions. Interestingly, while enteroviruses use the proteolytic activity of their 2A protein to degrade FG-nucleoporins, cardioviruses act by triggering phosphorylation of these proteins by cellular kinases. By targeting the nuclear pore complex, picornaviruses recruit nuclear proteins to the cytoplasm, where they increase viral genome translation and replication; they affect nuclear translocation of cytoplasmic proteins such as transcription factors that induce innate immune responses and retain host mRNA in the nucleus thereby preventing cell emergency responses and likely making the ribosomal machinery available for translation of viral RNAs.
Keywords: 2A protease; 3C protease; RAN GTPase; karyopherin; leader (L) protein; nuclear pore complex; nucleoporins; picornavirus.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

