Vaccines for Protecting Infants from Bacterial Causes of Diarrheal Disease
- PMID: 34202102
- PMCID: PMC8303436
- DOI: 10.3390/microorganisms9071382
Vaccines for Protecting Infants from Bacterial Causes of Diarrheal Disease
Abstract
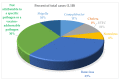
The global diarrheal disease burden for Shigella, enterotoxigenic Escherichia coli (ETEC), and Campylobacter is estimated to be 88M, 75M, and 75M cases annually, respectively. A vaccine against this target trio of enteric pathogens could address about one-third of diarrhea cases in children. All three of these pathogens contribute to growth stunting and have demonstrated increasing resistance to antimicrobial agents. Several combinations of antigens are now recognized that could be effective for inducing protective immunity against each of the three target pathogens in a single vaccine for oral administration or parenteral injection. The vaccine combinations proposed here would result in a final product consistent with the World Health Organization's (WHO) preferred product characteristics for ETEC and Shigella vaccines, and improve the vaccine prospects for support from Gavi, the Vaccine Alliance, and widespread uptake by low- and middle-income countries' (LMIC) public health stakeholders. Broadly protective antigens will enable multi-pathogen vaccines to be efficiently developed and cost-effective. This review describes how emerging discoveries for each pathogen component of the target trio could be used to make vaccines, which could help reduce a major cause of poor health, reduced cognitive development, lost economic productivity, and poverty in many parts of the world.
Keywords: Campylobacter vaccine; ETEC vaccine; Shigella vaccine; adjuvants; disease burden; models of disease; mucosal immunity; multi-pathogen enteric vaccines; stunting.
Conflict of interest statement
The authors declare no conflict of interest. R.W.K. is an inventor on several patents associated with Invaplex technology which are held by the US Government.
Figures

Similar articles
-
Potential impact and cost-effectiveness of future ETEC and Shigella vaccines in 79 low- and lower middle-income countries.Vaccine X. 2019 Apr 18;2:100024. doi: 10.1016/j.jvacx.2019.100024. eCollection 2019 Aug 9. Vaccine X. 2019. PMID: 31384741 Free PMC article.
-
Considerations for development of whole cell bacterial vaccines to prevent diarrheal diseases in children in developing countries.Vaccine. 2005 May 16;23(26):3369-85. doi: 10.1016/j.vaccine.2004.12.029. Vaccine. 2005. PMID: 15837361 Review.
-
Status of vaccine research and development for enterotoxigenic Escherichia coli.Vaccine. 2016 Jun 3;34(26):2880-2886. doi: 10.1016/j.vaccine.2016.02.076. Epub 2016 Mar 15. Vaccine. 2016. PMID: 26988259
-
An assessment of enterotoxigenic Escherichia coli and Shigella vaccine candidates for infants and children.Vaccine. 2015 Feb 18;33(8):954-65. doi: 10.1016/j.vaccine.2014.11.049. Epub 2014 Dec 5. Vaccine. 2015. PMID: 25482842 Review.
-
Vaccines against Shigella and enterotoxigenic Escherichia coli: A summary of the 2016 VASE Conference.Vaccine. 2017 Dec 14;35(49 Pt A):6775-6782. doi: 10.1016/j.vaccine.2017.09.045. Epub 2017 Oct 4. Vaccine. 2017. PMID: 28987444
Cited by
-
Anti-Toxin Responses to Natural Enterotoxigenic Escherichia coli (ETEC) Infection in Adults and Children in Bangladesh.Microorganisms. 2023 Oct 9;11(10):2524. doi: 10.3390/microorganisms11102524. Microorganisms. 2023. PMID: 37894182 Free PMC article.
-
Heat-Stable Enterotoxin Secretions Assessed via ICP-MS Reveal Iron-Mediated Regulation of Virulence in CFA/I- and CS6-Expressing ETEC Isolates.Cells. 2023 Feb 10;12(4):567. doi: 10.3390/cells12040567. Cells. 2023. PMID: 36831233 Free PMC article.
-
Efficient production of immunologically active Shigella invasion plasmid antigens IpaB and IpaH using a cell-free expression system.Appl Microbiol Biotechnol. 2022 Jan;106(1):401-414. doi: 10.1007/s00253-021-11701-4. Epub 2021 Dec 21. Appl Microbiol Biotechnol. 2022. PMID: 34932164 Free PMC article.
-
Conserved antigens for enteric vaccines.Vaccine. 2025 Mar 19;50:126828. doi: 10.1016/j.vaccine.2025.126828. Epub 2025 Feb 5. Vaccine. 2025. PMID: 39914256 Free PMC article. Review.
-
Repeat modules and N-linked glycans define structure and antigenicity of a critical enterotoxigenic E. coli adhesin.PLoS Pathog. 2024 Sep 16;20(9):e1012241. doi: 10.1371/journal.ppat.1012241. eCollection 2024 Sep. PLoS Pathog. 2024. PMID: 39283948 Free PMC article.
References
-
- Kotloff K.L., Nataro J.P., Blackwelder W.C., Nasrin D., Farag T.H., Panchalingam S., Wu Y., Sow S.O., Sur D., Breiman R.F., et al. Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the Global Enteric Multicenter Study, GEMS): A prospective, case-control study. Lancet. 2013;382:209–222. doi: 10.1016/S0140-6736(13)60844-2. - DOI - PubMed
-
- Liu J., Platts-Mills J.A., Juma J., Kabir F., Nkeze J., Okoi C., Operario D.J., Uddin J., Ahmed S., Alonso P.L., et al. Use of quantitative molecular diagnostic methods to identify causes of diarrhoea in children: A reanalysis of the GEMS case-control study. Lancet. 2016;388:1291–1301. doi: 10.1016/S0140-6736(16)31529-X. - DOI - PMC - PubMed
-
- Platts-Mills J.A., Liu J., Rogawski E.T., Kabir F., Lertsethtakarn P., Siguas M., Khan S.S., Praharaj I., Murei A., Nshama R., et al. Use of quantitative molecular diagnostic methods to assess the aetiology, burden, and clinical characteristics of diarrhoea in children in low-resource settings: A reanalysis of the MAL-ED cohort study. Lancet Glob. Health. 2018;6:e1309–e1318. doi: 10.1016/S2214-109X(18)30349-8. - DOI - PMC - PubMed
-
- GBD 2016 Diarrhoeal Disease Collaborators Estimates of the global, regional, and national morbidity, mortality, and aetiologies of diarrhoea in 195 countries: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect. Dis. 2018;18:1211–1228. doi: 10.1016/S1473-3099(18)30362-1. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

