Modulation of Stem Cell Progeny by Probiotics during Regeneration of Gastric Mucosal Erosions
- PMID: 34203400
- PMCID: PMC8301058
- DOI: 10.3390/biology10070596
Modulation of Stem Cell Progeny by Probiotics during Regeneration of Gastric Mucosal Erosions
Abstract
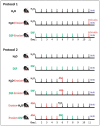
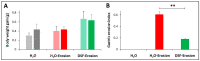
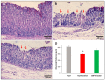
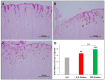
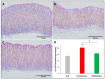
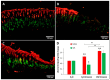
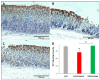
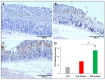
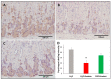
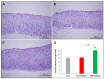
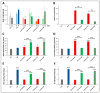
Patients with gastric mucosal erosions are predisposed to chronic gastritis, ulcer or even cancer. The repair of mucosal erosions involves several events including proliferation of gastric epithelial stem cells. The aim of this study was to investigate the effects of the probiotic mixture of De Simone Formulation on gastric epithelial stem cell lineages in mouse models of gastric mucosal erosions. Gastric erosions were induced by a single oral gavage of 80% ethanol containing 15 mg/mL acetylsalicylic acid (5 mL/kg) following a daily dose of probiotic mixture (5 mg/day/mouse) for 10 days. In another protocol, erosions were induced by a daily gavage of acetylsalicylic acid (400 mg/kg/day/mouse) for 5 days before or after daily administration of probiotic mixture for 5 days. Control mice received water gavage for 10 days. All mice were injected with bromodeoxyuridine two hours before sacrifice to label S-phase cells. The stomachs of all mice were processed for histological examination, lectin binding, and immunohistochemical analysis. The results reveal that mice that received probiotics before or after the induction of erosion showed a decrease in erosion index with an increase in gastric epithelial stem/progenitor cell proliferation and enhanced production of mucus, trefoil factors, and ghrelin by mucous and enteroendocrine cell lineages. These mice also showed restoration of the amount of H+,K+-ATPase and pepsinogen involved in the production of the harsh acidic environment by parietal and chief cell lineages. In conclusion, this study demonstrates the beneficial effects of probiotics against gastric mucosal erosion and highlights the involvement and modulation of proliferative stem cells and their multiple glandular epithelial cell lineages.
Keywords: H+,K+-ATPase; cell proliferation; gastric erosion; mucus; probiotics; trefoil factor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Probiotics Upregulate Trefoil Factors and Downregulate Pepsinogen in the Mouse Stomach.Int J Mol Sci. 2019 Aug 10;20(16):3901. doi: 10.3390/ijms20163901. Int J Mol Sci. 2019. PMID: 31405107 Free PMC article.
-
Preventive effect of probiotics and α-tocopherol on ethanol-induced gastric mucosal injury in rats.J Med Food. 2011 Jan-Feb;14(1-2):173-9. doi: 10.1089/jmf.2010.0040. J Med Food. 2011. PMID: 21244242
-
Simian virus 40 T antigen-induced amplification of pre-parietal cells in transgenic mice. Effects on other gastric epithelial cell lineages and evidence for a p53-independent apoptotic mechanism that operates in a committed progenitor.J Biol Chem. 1995 Jun 30;270(26):15777-88. doi: 10.1074/jbc.270.26.15777. J Biol Chem. 1995. PMID: 7797580
-
Effect of probiotics on aspirin-induced gastric mucosal lesions.Turk J Gastroenterol. 2011 Feb;22(1):18-26. doi: 10.4318/tjg.2011.0151. Turk J Gastroenterol. 2011. PMID: 21480106
-
Capsaicin as new orally applicable gastroprotective and therapeutic drug alone or in combination with nonsteroidal anti-inflammatory drugs in healthy human subjects and in patients.Prog Drug Res. 2014;68:209-58. doi: 10.1007/978-3-0348-0828-6_9. Prog Drug Res. 2014. PMID: 24941671 Review.
Cited by
-
Changes in the salivary metabolome in patients with chronic erosive gastritis.BMC Gastroenterol. 2023 May 19;23(1):161. doi: 10.1186/s12876-023-02803-6. BMC Gastroenterol. 2023. PMID: 37208605 Free PMC article.
-
A systematic review on selection characterization and implementation of probiotics in human health.Food Sci Biotechnol. 2023 Jan 10;32(4):423-440. doi: 10.1007/s10068-022-01210-z. eCollection 2023 Mar. Food Sci Biotechnol. 2023. PMID: 36911328 Free PMC article. Review.
References
Grants and funding
LinkOut - more resources
Full Text Sources

