Effective Tumor Targeting by EphA2-Agonist-Biotin-Streptavidin Conjugates
- PMID: 34204178
- PMCID: PMC8235110
- DOI: 10.3390/molecules26123687
Effective Tumor Targeting by EphA2-Agonist-Biotin-Streptavidin Conjugates
Abstract
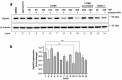
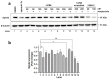
We recently reported on a potent synthetic agent, 135H11, that selectively targets the receptor tyrosine kinase, EphA2. While 135H11 possesses a relatively high binding affinity for the ligand-binding domain of EphA2 (Kd~130 nM), receptor activation in the cell required the synthesis of dimeric versions of such agent (namely 135H12). This was expected given that the natural ephrin ligands also need to be dimerized or clustered to elicit agonistic activity in cell. In the present report we investigated whether the agonistic activity of 135H11 could be enhanced by biotin conjugation followed by complex formation with streptavidin. Therefore, we measured the agonistic EphA2 activity of 135H11-biotin (147B5) at various agent/streptavidin ratios, side by side with 135H12, and a scrambled version of 147B5 in pancreatic- and breast-cancer cell lines. The (147B5)n-streptavidin complexes (when n = 2, 3, 4, but not when n = 1) induced a strong receptor degradation effect in both cell lines compared to 135H12 or the (scrambled-147B5)4-streptavidin complex as a control, indicating that multimerization of the targeting agent resulted in an increased ability to cause receptor clustering and internalization. Subsequently, we prepared an Alexa-Fluor-streptavidin conjugate to demonstrate that (147B5)4-AF-streptavidin, but not the scrambled equivalent complex, concentrates in pancreatic and breast cancers in orthotopic nude-mouse models. Hence, we conclude that these novel targeting agents, with proper derivatization with imaging reagents or chemotherapy, can be used as diagnostics, and/or to deliver chemotherapy selectively to EphA2-expressing tumors.
Keywords: agonistic EphA2 peptides; breast cancer; cancer imaging; orthotopic cancer models; pancreatic cancer; streptavidin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Targefrin: A Potent Agent Targeting the Ligand Binding Domain of EphA2.J Med Chem. 2022 Nov 24;65(22):15443-15456. doi: 10.1021/acs.jmedchem.2c01391. Epub 2022 Nov 4. J Med Chem. 2022. PMID: 36331527 Free PMC article.
-
Structure-Based Design of Novel EphA2 Agonistic Agents with Nanomolar Affinity in Vitro and in Cell.ACS Chem Biol. 2018 Sep 21;13(9):2633-2644. doi: 10.1021/acschembio.8b00556. Epub 2018 Aug 29. ACS Chem Biol. 2018. PMID: 30110533 Free PMC article.
-
Therapeutic Targeting of Pancreatic Cancer via EphA2 Dimeric Agonistic Agents.Pharmaceuticals (Basel). 2020 May 10;13(5):90. doi: 10.3390/ph13050090. Pharmaceuticals (Basel). 2020. PMID: 32397624 Free PMC article.
-
Reduction of Circulating Cancer Cells and Metastases in Breast-Cancer Models by a Potent EphA2-Agonistic Peptide-Drug Conjugate.J Med Chem. 2018 Mar 8;61(5):2052-2061. doi: 10.1021/acs.jmedchem.7b01837. Epub 2018 Feb 27. J Med Chem. 2018. PMID: 29470068 Free PMC article.
-
Streptavidin-biotin binding energetics.Biomol Eng. 1999 Dec 31;16(1-4):39-44. doi: 10.1016/s1050-3862(99)00042-x. Biomol Eng. 1999. PMID: 10796983 Review.
Cited by
-
EPHA2 Receptor as a Possible Therapeutic Target in Viral Infections.Curr Med Chem. 2024;31(35):5670-5701. doi: 10.2174/0109298673256638231003111234. Curr Med Chem. 2024. PMID: 37828671 Review.
-
Targefrin: A Potent Agent Targeting the Ligand Binding Domain of EphA2.J Med Chem. 2022 Nov 24;65(22):15443-15456. doi: 10.1021/acs.jmedchem.2c01391. Epub 2022 Nov 4. J Med Chem. 2022. PMID: 36331527 Free PMC article.
-
EPH/Ephrin-Targeting Treatment in Breast Cancer: A New Chapter in Breast Cancer Therapy.Int J Mol Sci. 2022 Dec 3;23(23):15275. doi: 10.3390/ijms232315275. Int J Mol Sci. 2022. PMID: 36499598 Free PMC article. Review.
-
Bio-SS-TS as a Targeted Antitumor Drug Exerts an Anti-Liver Cancer Effect by Enhancing Mitochondria-Dependent Apoptosis.Biol Proced Online. 2025 Mar 28;27(1):11. doi: 10.1186/s12575-025-00272-7. Biol Proced Online. 2025. PMID: 40155811 Free PMC article.
References
-
- Walker-Daniels J., Coffman K., Azimi M., Rhim J.S., Bostwick D.G., Snyder P., Kerns B.J., Waters D.J., Kinch M.S. Overexpression of the EphA2 tyrosine kinase in prostate cancer. Prostate. 1999;41:275–280. doi: 10.1002/(SICI)1097-0045(19991201)41:4275::AID-PROS83.0.CO;2-T. - DOI - PubMed
-
- Zelinski D.P., Zantek N.D., Stewart J.C., Irizarry A.R., Kinch M.S. EphA2 overexpression causes tumorigenesis of mammary epithelial cells. Cancer Res. 2001;61:2301–2306. - PubMed
-
- Coffman K.T., Hu M., Carles-Kinch K., Tice D., Donacki N., Munyon K., Kifle G., Woods R., Langermann S., Kiener P.A., et al. Differential EphA2 epitope display on normal versus malignant cells. Cancer Res. 2003;63:7907–7912. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

