The Role of Capsid in the Early Steps of HIV-1 Infection: New Insights into the Core of the Matter
- PMID: 34204384
- PMCID: PMC8234406
- DOI: 10.3390/v13061161
The Role of Capsid in the Early Steps of HIV-1 Infection: New Insights into the Core of the Matter
Abstract
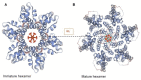
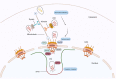
In recent years, major advances in research and experimental approaches have significantly increased our knowledge on the role of the HIV-1 capsid in the virus life cycle, from reverse transcription to integration and gene expression. This makes the capsid protein a good pharmacological target to inhibit HIV-1 replication. This review covers our current understanding of the role of the viral capsid in the HIV-1 life cycle and its interaction with different host factors that enable reverse transcription, trafficking towards the nucleus, nuclear import and integration into host chromosomes. It also describes different promising small molecules, some of them in clinical trials, as potential targets for HIV-1 therapy.
Keywords: HIV-1; capsid; drugs; integration; nuclear entry; reverse transcription.
Conflict of interest statement
The authors declare no conflict of interests.
Figures
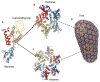
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

