Malnutrition Aggravates Alterations Observed in the Gut Structure and Immune Response of Mice Infected with Leishmania infantum
- PMID: 34207946
- PMCID: PMC8230684
- DOI: 10.3390/microorganisms9061270
Malnutrition Aggravates Alterations Observed in the Gut Structure and Immune Response of Mice Infected with Leishmania infantum
Abstract
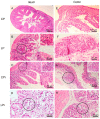
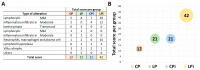
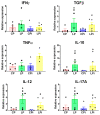
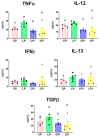
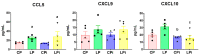
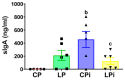
Malnutrition is a risk factor for developing visceral leishmaniasis and its severe forms. Our group demonstrated that malnourished animals infected with Leishmania infantum had severe atrophies in lymphoid organs and T cell subpopulations as well as altered levels of thymic and splenic chemotactic factors, all of which resulted in dysfunctional lymphoid microenvironments that promoted parasite proliferation. Here, we hypothesize that malnutrition preceding parasite infection leads to structural and immunological changes in the gut mucosae, resulting in a failure in the immune response sensed in the intestine. To evaluate this, we analyzed the immunopathological events resulting from protein malnutrition in the guts of BALB/c mice infected with L. infantum. We observed lymphocytic/lymphoplasmacytic inflammatory infiltrates and lymphoid hyperplasia in the duodenum of well-nourished-infected mice; such alterations were worsened when malnutrition preceded infection. Parasite infection induced a significant increase of duodenal immunoglobulin A (IgA) of well-nourished animals, but those levels were significantly decreased in malnourished-infected mice. In addition, increased levels of Th17-related cytokines in duodenums of malnourished animals supported local inflammation. Together, our results suggest that the gut plays a potential role in responses to L. infantum infection-and that such responses are impaired in malnourished individuals.
Keywords: IgA; Leishmania infantum; duodenum; gut; inflammation; malnutrition; visceral leishmaniasis.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures



References
Grants and funding
- P.C. Grant # VPPCB-007-FIO-18-2-74/FIOCRUZ - INOVA Program
- P.C. Emergentes grant # E-26/010.002168/2019/Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro
- P.C. JCNE E-26/201.545/2014 and JCNE E-26/203.253/2017/Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro
- Sediadas grant # E-26/010.101083/2018/Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro
- P.C. Universal grant No. 448007/2014-2/Conselho Nacional de Desenvolvimento Científico e Tecnológico
LinkOut - more resources
Full Text Sources
Miscellaneous

