The Role of Traditional Chinese Formula Ding-Kun Pill (DKP) in Expected Poor Ovarian Response Women (POSEIDON Group 4) Undergoing In Vitro Fertilization-Embryo Transfer: A Multicenter, Randomized, Double-Blind, Placebo-Controlled Trial
- PMID: 34220712
- PMCID: PMC8247913
- DOI: 10.3389/fendo.2021.675997
The Role of Traditional Chinese Formula Ding-Kun Pill (DKP) in Expected Poor Ovarian Response Women (POSEIDON Group 4) Undergoing In Vitro Fertilization-Embryo Transfer: A Multicenter, Randomized, Double-Blind, Placebo-Controlled Trial
Abstract
Objective: The primary objective of the study was to assess traditional Chinese formula DKP supplementation in terms of efficacy and safety on reproductive outcomes of expected poor ovarian responder (POR, POSEIDON Group 4) undergoing in vitro fertilization-embryo transfer (IVF-ET).
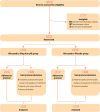
Design setting and participants: Women eligible for IVF-ET were invited to participate in this randomized, double-blind, placebo-controlled, superiority trial at academic fertility centers of ten public hospitals in Chinese Mainland. A total of 462 patients (35-44 years) equally divided between DKP and placebo groups with antral follicle count (AFC) <5 or anti-müllerian hormone (AMH) <1.2 ng/ml were randomized.
Interventions: All participants were given DKP or 7 g placebo twice daily on the previous menstrual cycle day 5 until oocyte retrieval, which took approximately 5 to 6 weeks.
Main outcome measure: The primary outcome was the ongoing pregnancy defined as more than 20 gestational weeks of an intrauterine living fetus confirmed by pelvic ultrasonography.
Results: Demographic characteristics were equally distributed between the study populations. Intention-to-treat (ITT) analysis revealed that ongoing pregnancy rate (OPR) was not significantly different between DKP and placebo groups [26.4% (61/231) versus 24.2% (56/231); relative risk (RR) 1.09, 95% confidence interval (CI) 0.80 to 1.49, P = 0.593]. No significant differences between groups were observed for the secondary outcomes. The additional per protocol (PP) analysis was in line with ITT results: OPR in DKP group was 27.2% (61/224) versus 24.1% (55/228) in placebo group [RR 1.13, 95%CI (0.82 to 1.55), P = 0.449]. After subgroup analysis the findings concluded that POR population of 35-37 years had a significantly higher OPR after 5-6 weeks of oral DKP (41.8%, 33/79) versus placebo (25.4%, 18/71) [RR 1.65, 95% CI (1.02 to 2.65), P = 0.034, P for interaction = 0.028].
Conclusion: This well-designed randomized controlled trial (RCT) offers new high-quality evidence to supplement existing retrospective literature concerning DKP performance in expected PORs. DKP could be recommended as a safe and natural remedy for expected PORs (aged 35-37 years) who fulfill the POSEIDON group 4 criteria. However, additional interventional clinical studies are undoubtedly required to be conducted in the future to validate this hypothesis.
Clinical trial registration: www.chictr.org.cn, identifier ChiCTR1900026614.
Keywords: Ding-Kun Pill; POSEIDON criteria; in vitro fertilization-embryo transfer; low prognosis; poor ovarian response; traditional Chinese medicine.
Copyright © 2021 Song, Gao, Cao, Xiang, Chen, Teng, Li, Liu, Wang, Zhang, Xu, Zhou, Huang and Sun.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Adamson GD, de Mouzon J, Chambers GM, Zegers-Hochschild F, Mansour R, Ishihara O, et al. International Committee for Monitoring Assisted Reproductive Technology: World Report on Assisted Reproductive Technology, 2011. Fertil Steril (2018) 110(6):1067–80. 10.1016/j.fertnstert.2018.06.039 - DOI - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

