Quantitative Assessment of Experimental Ocular Inflammatory Disease
- PMID: 34220797
- PMCID: PMC8250853
- DOI: 10.3389/fimmu.2021.630022
Quantitative Assessment of Experimental Ocular Inflammatory Disease
Abstract
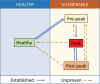
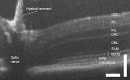
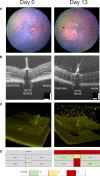
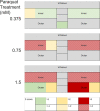
Ocular inflammation imposes a high medical burden on patients and substantial costs on the health-care systems that mange these often chronic and debilitating diseases. Many clinical phenotypes are recognized and classifying the severity of inflammation in an eye with uveitis is an ongoing challenge. With the widespread application of optical coherence tomography in the clinic has come the impetus for more robust methods to compare disease between different patients and different treatment centers. Models can recapitulate many of the features seen in the clinic, but until recently the quality of imaging available has lagged that applied in humans. In the model experimental autoimmune uveitis (EAU), we highlight three linked clinical states that produce retinal vulnerability to inflammation, all different from healthy tissue, but distinct from each other. Deploying longitudinal, multimodal imaging approaches can be coupled to analysis in the tissue of changes in architecture, cell content and function. This can enrich our understanding of pathology, increase the sensitivity with which the impacts of therapeutic interventions are assessed and address questions of tissue regeneration and repair. Modern image processing, including the application of artificial intelligence, in the context of such models of disease can lay a foundation for new approaches to monitoring tissue health.
Keywords: EAU; OCT; automated analysis; image processing; uveitis.
Copyright © 2021 Bradley, Ward, Hsue, Liu, Copland, Dick and Nicholson.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Caspi RR, Roberge FG, Chan CC, Wiggert B, Chader GJ, Rozenszajn LA, et al. . A New Model of Autoimmune Disease. Experimental Autoimmune Uveoretinitis Induced in Mice With Two Different Retinal Antigens. J Immunol (1988) 140(5):1490–5. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

