A randomized phase 1b cross-over study of the safety of low-dose pioglitazone for treatment of autosomal dominant polycystic kidney disease
- PMID: 34221381
- PMCID: PMC8243264
- DOI: 10.1093/ckj/sfaa232
A randomized phase 1b cross-over study of the safety of low-dose pioglitazone for treatment of autosomal dominant polycystic kidney disease
Abstract
Background: Autosomal dominant polycystic kidney disease (ADPKD) is one of the most common monogenetic disorders in humans and is characterized by numerous fluid-filled cysts that grow slowly, resulting in end-stage renal disease in the majority of patients. Preclinical studies have indicated that treatment with low-dose thiazolidinediones, such as pioglitazone, decrease cyst growth in rodent models of PKD.
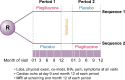
Methods: This Phase 1b cross-over study compared the safety of treatment with a low dose (15 mg) of the peroxisome proliferator-activated receptor-γ (PPAR-γ) agonist pioglitazone or placebo in PKD patients, with each treatment given for 1 year. The study monitored known side effects of PPAR-γ agonist treatment, including fluid retention and edema. Liver enzymes and risk of hypoglycemia were assessed throughout the study. As a secondary objective, the efficacy of low-dose pioglitazone was followed using a primary assessment of total kidney volume (TKV), blood pressure (BP) and kidney function.
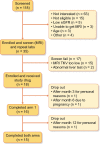
Results: Eighteen patients were randomized and 15 completed both arms. Compared with placebo, allocation to pioglitazone resulted in a significant decrease in total body water as assessed by bioimpedance analysis {mean difference 0.16 Ω [95% confidence interval (CI) 0.24-2.96], P = 0.024} and no differences in episodes of heart failure, clinical edema or change in echocardiography. Allocation to pioglitazone led to no difference in the percent change in TKV of -3.5% (95% CI -8.4-1.4, P = 0.14), diastolic BP and microalbumin:creatinine ratio.
Conclusions: In this small pilot trial in people with ADPKD but without diabetes, pioglitazone 15 mg was found to be as safe as placebo. Larger and longer-term randomized trials powered to assess effects on TKV are needed.
Keywords: MRI; PPARγ; agonists; anti-hypertensive; crossover design; total kidney volume.
© The Author(s) 2021. Published by Oxford University Press on behalf of ERA-EDTA.
Figures



References
-
- Schrier RW, McFann KK, Johnson AM.. Epidemiological study of kidney survival in autosomal dominant polycystic kidney disease. Kidney Int 2003; 63: 678–685 - PubMed
-
- Harris PC, Hopp K.. The mutation, a key determinant of phenotype in ADPKD. J Am Soc Nephrol 2013; 24: 868–870 - PubMed
-
- Hanaoka K, Qian F, Boletta A. et al. Co-assembly of polycystin-1 and -2 produces unique cation-permeable currents. Nature 2000; 408: 990–994 - PubMed
-
- Hanaoka K, Guggino WB.. cAMP regulates cell proliferation and cyst formation in autosomal polycystic kidney disease cells. J Am Soc Nephrol 2000; 11: 1179–1187 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

