The bromodomain inhibitor JQ1+ reduces calcium-sensing receptor activity in pituitary cell lines
- PMID: 34223822
- PMCID: PMC8345903
- DOI: 10.1530/JME-21-0030
The bromodomain inhibitor JQ1+ reduces calcium-sensing receptor activity in pituitary cell lines
Abstract
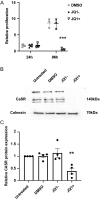
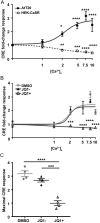
Corticotrophinomas represent 10% of all surgically removed pituitary adenomas, however, current treatment options are often not effective, and there is a need for improved pharmacological treatments. Recently, JQ1+, a bromodomain inhibitor that promotes gene transcription by binding acetylated histone residues and recruiting transcriptional machinery, has been shown to reduce proliferation in a murine corticotroph cell line, AtT20. RNA-Seq analysis of AtT20 cells following treatment with JQ1+ identified the calcium-sensing receptor (CaSR) gene as significantly downregulated, which was subsequently confirmed using real-time PCR and Western blot analysis. CaSR is a G protein-coupled receptor that plays a central role in calcium homeostasis but can elicit non-calcitropic effects in multiple tissues, including the anterior pituitary where it helps regulate hormone secretion. However, in AtT20 cells, CaSR activates a tumour-specific cAMP pathway that promotes ACTH and PTHrP hypersecretion. We hypothesised that the Casr promoter may harbour binding sites for BET proteins, and using chromatin immunoprecipitation (ChIP)-sequencing demonstrated that the BET protein Brd3 binds to the promoter of the Casr gene. Assessment of CaSR signalling showed that JQ1+ significantly reduced Ca2+e-mediated increases in intracellular calcium (Ca2+i) mobilisation and cAMP signalling. However, the CaSR-negative allosteric modulator, NPS-2143, was unable to reduce AtT20 cell proliferation, indicating that reducing CaSR expression rather than activity is likely required to reduce pituitary cell proliferation. Thus, these studies demonstrate that reducing CaSR expression may be a viable option in the treatment of pituitary tumours. Moreover, current strategies to reduce CaSR activity, rather than protein expression for cancer treatments, may be ineffective.
Keywords: G protein-coupled receptor; corticotrophinoma; epigenetic modification; pituitary tumourigenesis.
Figures



Similar articles
-
Effects of epigenetic pathway inhibitors on corticotroph tumour AtT20 cells.Endocr Relat Cancer. 2020 Mar;27(3):163-174. doi: 10.1530/ERC-19-0448. Endocr Relat Cancer. 2020. PMID: 31935194 Free PMC article.
-
The calcium-sensing receptor couples to Galpha(s) and regulates PTHrP and ACTH secretion in pituitary cells.J Endocrinol. 2010 Mar;204(3):287-97. doi: 10.1677/JOE-09-0183. Epub 2009 Dec 23. J Endocrinol. 2010. PMID: 20032198 Free PMC article.
-
Calcium-sensing receptor expression and signalling in human parathyroid adenomas and primary hyperplasia.Clin Endocrinol (Oxf). 2000 Mar;52(3):339-48. doi: 10.1046/j.1365-2265.2000.00933.x. Clin Endocrinol (Oxf). 2000. PMID: 10718832
-
The calcium-sensing receptor and the hallmarks of cancer.Biochim Biophys Acta. 2016 Jun;1863(6 Pt B):1398-407. doi: 10.1016/j.bbamcr.2015.11.017. Epub 2015 Nov 30. Biochim Biophys Acta. 2016. PMID: 26608608 Review.
-
Insights into calcium-sensing receptor trafficking and biased signalling by studies of calcium homeostasis.J Mol Endocrinol. 2018 Jul;61(1):R1-R12. doi: 10.1530/JME-18-0049. Epub 2018 Mar 29. J Mol Endocrinol. 2018. PMID: 29599414 Review.
Cited by
-
Calcium sensing receptor expression is downregulated in gastroenteropancreatic neuroendocrine tumours via epigenetic mechanisms.Int J Cancer. 2025 Mar 1;156(5):980-992. doi: 10.1002/ijc.35264. Epub 2024 Nov 23. Int J Cancer. 2025. PMID: 39579056 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

