[Determination of 16 organophosphate esters in human blood by high performance liquid chromatography-tandem mass spectrometry combined with liquid-liquid extraction and solid phase extraction]
- PMID: 34227360
- PMCID: PMC9274832
- DOI: 10.3724/SP.J.1123.2020.07033
[Determination of 16 organophosphate esters in human blood by high performance liquid chromatography-tandem mass spectrometry combined with liquid-liquid extraction and solid phase extraction]
Abstract
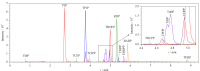
Measurement of organophosphate esters (OPEs) in human body fluids is important for understanding human internal exposure to OPEs and for assessing related health risks. Most of the current studies have focused on the determination of OPE metabolites in human urine, as OPEs are readily metabolized into their diester or hydroxylated forms in the human body. However, given the existence of one metabolite across multiple OPEs or multiple metabolites of one OPE, as well as the low metabolic rates of several OPEs in in vitro studies, the reliability of urinary OPE metabolites as biomarkers for specific OPEs is needs to be treated with caution.Human blood is a matrix that is in contact with all body organs and tissues, and the blood levels of compounds may better represent the doses that reach target tissues. Currently, only a few studies have investigated the occurrence of OPEs in human blood by different analytical methods, and the variety of OPEs considered is limited. In this study, a method based on liquid chromatography-tandem mass spectrometry (LC-MS/MS) was developed for the simultaneous determination of 16 OPEs in human blood, and the extraction efficiency of the solid phase extraction (SPE) column for OPEs was verified. To human blood samples, 10 ng of an internal standard was added, followed by mixing and aging for 30 min. The samples were extracted three times with acetonitrile using a shaker, and then purified on ENVI-18 cartridges with acetonitrile containing 25% dichloromethane as the eluent. Finally, the OPEs were analyzed by high performance liquid chromatography-tandem mass spectrometry. After optimization of the analytical column and mobile phases, the analytes were separated on a BEH C18 column (100 mm×2.1 mm, 1.7 μm) by gradient elution using methanol and 5 mmol/L ammonium acetate in water as the mobile phase. Then, the analytes were ionized in electrospray ionization positive (ESI+) mode and detected in the multiple reaction monitoring (MRM) mode. The mass spectral parameters, including the precursor ion, product ion, declustering potential, entrance potential, and collision cell exit potential, were optimized. The results were quantified by the internal standard method. The limits of detection (LOD, S/N=3) of the OPEs were in the range of 0.0038-0.882 ng/mL. The calibration curves for the 16 OPEs showed good linear relationships in the range of 0.1-50 ng/mL, and the correlation coefficients were >0.995. The extraction efficiency of the ENVI-18 column for the 16 OPEs was validated, and the average recoveries of the target compounds were 54.6%-104%. The average recoveries (n=3) of 15 OPEs, except trimethyl phosphate (TMP), in whole blood at three spiked levels were in the range of 53.1%-126%, and the relative standard deviations (RSDs) were in the range of 0.15%-12.6%. The average recoveries of six internal standards were in the range of 66.8%-91.6% except for TMP-d9 (39.1%), with RSDs of 3.52%-6.85%. The average matrix effects of the OPEs in whole blood were 56.4%-103.0%. Significant matrix effects were found for resorcinol bis(diphenyl phosphate) (RDP) (75.8%±1.4%), trimethylphenyl phosphate (TMPP) (68.4%±1.0%), 2-ethylhexyl di-phenyl phosphate (EHDPP) (56.4%±12.4%), and bisphenol-A bis(diphenyl phosphate) (BABP) (58.5%±0.4%). However, these effects could be corrected by similar signal suppressions of the corresponding internal standard (TPHP-d15, 77.4%±7.5%). This method is simple, highly sensitive, and suitable for the determination of OPEs in human blood. Fifteen human whole blood samples were collected to quantify the 16 OPEs using the developed method. The total concentrations of the OPEs ranged from 1.50 to 7.99 ng/mL. The detection frequencies of eight OPEs were higher than 50%. Tri-iso-butyl phosphate (TiBP), tri(2-chloroethyl) phosphate (TCEP), and tri(1-chloro-2-propyl) phosphate (TCIPP) were the dominant OPEs, with median concentrations of 0.813, 0.764, and 0.690 ng/mL, respectively. These results indicated widespread human exposure to OPEs, which should be of concern.
人体体液中有机磷酸酯(OPEs)浓度的测定对于了解人体OPEs的暴露水平以及评估人体健康风险具有重要意义。然而,目前的研究大多数集中于尿液中OPEs代谢物含量的分析测定,将其作为人体OPEs暴露的生物标志物,而对人体血液中OPEs的分析研究较少,仅有的少量研究涉及的OPEs种类有限。该研究在优化前处理过程(固相萃取,SPE)和色谱分离的基础上,建立了人体血液中16种OPEs的超高效液相色谱-串联质谱(UPLC-MS-MS)测定方法。血液样品经过乙腈摇床萃取后,经ENVI-18 SPE小柱净化,然后采用Acquity UPLC BEH C18色谱柱,以甲醇/5 mmol/L的乙酸铵水溶液为流动相进行梯度洗脱对目标物进行分离,最后进行LC-MS/MS测定。质谱分析采用电喷雾正离子模式电离,多重反应监测模式测定,内标法定量。在优化的检测条件下,16种OPEs的检出限为0.0038~0.882 ng/mL。除磷酸三甲酯(TMP)外,其余15种OPEs在3个浓度水平的血液基质加标回收率为53.1%~126%,相对标准偏差为0.15%~12.6%。样品的基质效应检测发现,4种OPEs存在明显的基质抑制,选用合适的同位素内标进行定量,可以部分消除基质影响。该方法样品前处理简单,灵敏度高,适用于人体血液样品中OPEs阻燃剂的测定。15个人体血液样本分析结果表明,OPEs的总浓度范围为1.50~7.99 ng/mL,其中8种OPEs的检出率均高于50%,磷酸三异丁酯(TiBP)、磷酸三(2-氯乙基)酯(TCEP)和磷酸三(1-氯-2-丙基)磷酸酯(TCIPP)为主要的OPEs,表明人体存在较为普遍的OPEs暴露,应该引起关注。
Keywords: high performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS); human blood; liquid-liquid extraction (LLE); organophosphate ester; solid phase extraction (SPE).
Similar articles
-
[Determination of organophosphate diesters in facility vegetable soils using ultra-high performance liquid chromatography-electrostatic field orbitrap high resolution mass spectrometry].Se Pu. 2023 Jan;41(1):58-65. doi: 10.3724/SP.J.1123.2022.05002. Se Pu. 2023. PMID: 36633077 Free PMC article. Chinese.
-
[Determination of eight organophosphate esters in animal-derived foods by ultra performance liquid chromatography-tandem mass spectrometry].Se Pu. 2025 Apr 8;43(4):309-316. doi: 10.3724/SP.J.1123.2024.07010. Se Pu. 2025. PMID: 40133196 Free PMC article. Chinese.
-
[Determination of eight environmental phenols in human urine samples by high-throughput solid-phase extraction-ultra-performance liquid chromatography-tandem mass spectrometry].Se Pu. 2020 Dec 8;38(12):1456-1464. doi: 10.3724/SP.J.1123.2020.07021. Se Pu. 2020. PMID: 34213261 Chinese.
-
[Advances in the development of detection techniques for organophosphate ester flame retardants in food].Se Pu. 2020 Dec 8;38(12):1369-1380. doi: 10.3724/SP.J.1123.2020.03026. Se Pu. 2020. PMID: 34213251 Review. Chinese.
-
Human internal exposure to organophosphate esters: A short review of urinary monitoring on the basis of biological metabolism research.J Hazard Mater. 2021 Sep 15;418:126279. doi: 10.1016/j.jhazmat.2021.126279. Epub 2021 Jun 5. J Hazard Mater. 2021. PMID: 34329041 Review.
Cited by
-
[Determination of organophosphorus pesticides based on graphene oxide aerogel solid phase extraction column].Se Pu. 2022 Jan;40(1):10-16. doi: 10.3724/SP.J.1123.2021.03032. Se Pu. 2022. PMID: 34985211 Free PMC article. Chinese.
-
[High-throughput screening of 54 alternative plasticizers in sesame oil using gas chromatography-quadrupole time-of-flight mass spectrometry].Se Pu. 2023 Nov;41(11):1010-1020. doi: 10.3724/SP.J.1123.2023.08007. Se Pu. 2023. PMID: 37968820 Free PMC article. Chinese.
-
[Determination of organophosphate diesters in facility vegetable soils using ultra-high performance liquid chromatography-electrostatic field orbitrap high resolution mass spectrometry].Se Pu. 2023 Jan;41(1):58-65. doi: 10.3724/SP.J.1123.2022.05002. Se Pu. 2023. PMID: 36633077 Free PMC article. Chinese.
References
-
- Wei G L, Li D Q, Zhuo M N, et al.. Environ Pollut, 2015,196:29 - PubMed
-
- Wang Y, Hou M, Zhang Q, et al.. Environ Sci Technol, 2017,51(19):10991 - PubMed
-
- Zhang T, Bai X Y, Lu S Y, et al.. Environ Int, 2018,121(Pt 2):1363 - PubMed
-
- Suhring R, Diamond M L, Scheringer M, et al.. Environ Sci Technol, 2016,50(14):7409 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous