Duodenal imaging on the spotlight: from A to Z
- PMID: 34232417
- PMCID: PMC8263847
- DOI: 10.1186/s13244-021-01045-y
Duodenal imaging on the spotlight: from A to Z
Abstract
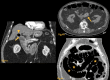
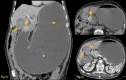
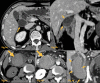
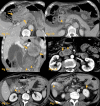
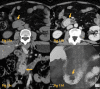
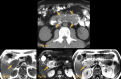
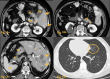
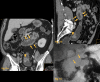
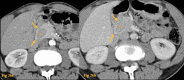
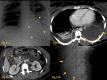
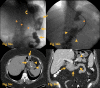
Abdominal computed tomography (CT) is frequently performed to evaluate gastrointestinal pathologic conditions. The majority of the gastrointestinal radiology literature has concentrated on the colon, stomach, and distal small bowel. The duodenum is often overlooked on imaging, namely on CT, but its anatomy (intra and retroperitoneal) and location in such close proximity to other viscera results in involvement by a multitude of primary and secondary processes, some of them exclusive to this bowel segment. While some conditions, like duplications, lipomas, and diverticula, are usually asymptomatic and are incidentalomas that have no pathologic significance, others are symptomatic and very relevant and should be recognized by every general radiologist: development conditions such as annular pancreas and gut malrotation; inflammatory processes such as ulcers and secondary involvement from pancreatitis; neoplastic conditions such as adenocarcinoma, lymphoma, or local extension from adjacent malignancies. They all can be reliably diagnosed with CT. In this article, we demonstrate the typical imaging features of various diseases involving the duodenum, such as developmental, traumatic, inflammatory, infectious, neoplastic, and postsurgical pathologic conditions in alphabetical order, focusing mainly on upper gastrointestinal series (UGIS) and CT but also some radiography, ultrasound, and magnetic resonance (MR) imaging.
Keywords: Abdomen; Computed tomography; Duodenum; Gastrointestinal; Magnetic resonance imaging; Ultrasound; Upper gastrointestinal series.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures








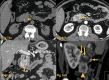
















References
-
- Lee CC, Ng WK, Lin KW, Lai TW, Li SM. Adenocarcinoma of the duodenum. Hong Kong Med J. 2008;14:67–69. - PubMed
-
- Lee EY, Andetta HB. Computed body tomography with MRI correlation. Alphen aan den Rijn: Wolters Kluwer; 2019.
Publication types
LinkOut - more resources
Full Text Sources

