Characterization of LRP4/Agrin Antibodies From a Patient With Myasthenia Gravis
- PMID: 34233932
- PMCID: PMC8448554
- DOI: 10.1212/WNL.0000000000012463
Characterization of LRP4/Agrin Antibodies From a Patient With Myasthenia Gravis
Abstract
Background and objective: To determine whether human anti-LRP4/agrin antibodies are pathogenic in mice and to investigate underpinning pathogenic mechanisms.
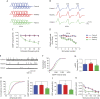
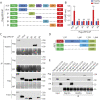
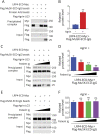
Methods: Immunoglobulin (Ig) was purified from a patient with myasthenia gravis (MG) with anti-LRP4/agrin antibodies and transferred to mice. Mice were characterized for body weight, muscle strength, twitch and tetanic force, neuromuscular junction (NMJ) functions including compound muscle action potential (CMAP) and endplate potentials, and NMJ structure. Effects of the antibodies on agrin-elicited muscle-specific tyrosine kinase (MuSK) activation and AChR clustering were studied and the epitopes of these antibodies were identified.
Results: Patient Ig-injected mice had MG symptoms, including weight loss and muscle weakness. Decreased CMAPs, reduced twitch and tetanus force, compromised neuromuscular transmission, and NMJ fragmentation and distortion were detected in patient Ig-injected mice. Patient Ig inhibited agrin-elicited MuSK activation and AChR clustering. The patient Ig recognized the β3 domain of LRP4 and the C-terminus of agrin and reduced agrin-enhanced LRP4-MuSK interaction.
Discussion: Anti-LRP4/agrin antibodies in the patient with MG is pathogenic. It impairs the NMJ by interrupting agrin-dependent LRP4-MuSK interaction.
© 2021 American Academy of Neurology.
Figures




Comment in
-
Antibodies to LRP4 and Agrin Are Pathogenic in Myasthenia Gravis: At the Junction Where It Happens.Neurology. 2021 Sep 7;97(10):463-464. doi: 10.1212/WNL.0000000000012471. Epub 2021 Jul 7. Neurology. 2021. PMID: 34233934 No abstract available.
Comment on
-
Antibodies to LRP4 and Agrin Are Pathogenic in Myasthenia Gravis: At the Junction Where It Happens.Neurology. 2021 Sep 7;97(10):463-464. doi: 10.1212/WNL.0000000000012471. Epub 2021 Jul 7. Neurology. 2021. PMID: 34233934 No abstract available.
References
-
- Gilhus NE, Tzartos S, Evoli A, Palace J, Burns TM, Verschuuren J. Myasthenia gravis. Nat Rev Dis Primers. 2019;5(1):30. - PubMed
-
- Li L, Xiong WC, Mei L. Neuromuscular junction formation, aging, and disorders. Annu Rev Physiol. 2018;80:159-188. - PubMed
-
- Gilhus NE, Verschuuren JJ. Myasthenia gravis: subgroup classification and therapeutic strategies. Lancet Neurol. 2015;14(10):1023-1036. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
