Ependymal cells-CSF flow regulates stress-induced depression
- PMID: 34234280
- PMCID: PMC8873010
- DOI: 10.1038/s41380-021-01202-1
Ependymal cells-CSF flow regulates stress-induced depression
Abstract
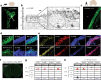
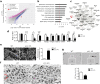
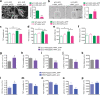
Major depressive disorder (MDD) is a severe, common mood disorder. While reduced cerebrospinal fluid (CSF) flow adversely affects brain metabolism and fluid balance in the aging population and during development, only indirect evidence links aberrant CSF circulation with many diseases including neurological, neurodegenerative, and psychiatric disorders, such as anxiety and depression. Here we show a very high concentration of p11 as a key molecular determinant for depression in ependymal cells, which is significantly decreased in patients with MDD, and in two mouse models of depression induced by chronic stress, such as restraint and social isolation. The loss of p11 in ependymal cells causes disoriented ependymal planar cell polarity (PCP), reduced CSF flow, and depression-like and anxiety-like behaviors. p11 intrinsically controls PCP core genes, which mediates CSF flow. Viral expression of p11 in ependymal cells specifically rescues the pathophysiological and behavioral deficits caused by loss of p11. Taken together, our results identify a new role and a key molecular determinant for ependymal cell-driven CSF flow in mood disorders and suggest a novel strategy for development of treatments for stress-associated neurological, neurodegenerative, and psychiatric disorders.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
-
- de Kloet ER, Joels M, Holsboer F. Stress and the brain: from adaptation to disease. Nat Rev Neurosci. 2005;6:463–75. - PubMed
-
- McEwen BS. Protective and damaging effects of stress mediators. N Engl J Med. 1998;338:171–9. - PubMed
-
- Pittenger C, Duman RS. Stress, depression, and neuroplasticity: a convergence of mechanisms. Neuropsychopharmacology. 2008;33:88–109. - PubMed
-
- Svenningsson P, Chergui K, Rachleff I, Flajolet M, Zhang X, El Yacoubi M, et al. Alterations in 5-HT1B receptor function by p11 in depression-like states. Science. 2006;311:77–80. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

