Repetitive Transcranial Magnetic Stimulation Improves Mild Cognitive Impairment Associated with Alzheimer's Disease in Mice by Modulating the miR-567/NEUROD2/PSD95 Axis
- PMID: 34239303
- PMCID: PMC8259939
- DOI: 10.2147/NDT.S311183
Repetitive Transcranial Magnetic Stimulation Improves Mild Cognitive Impairment Associated with Alzheimer's Disease in Mice by Modulating the miR-567/NEUROD2/PSD95 Axis
Abstract
Background: Mild cognitive impairment (MCI) is a typical symptom of early Alzheimer's disease (AD) and is driven by the dysfunction of microRNAs (miRs). Repetitive transcranial magnetic stimulation (rTMS) is a non-invasive technique for handling neuropsychiatric disorders and has universally effects on the functions of miRs. In the current study, the improvement effects of rTMS on MCI associated with AD were explored by focusing on miR-567/NEUROD2/PSD95 axis.
Methods: MCI was induced in mice using scopolamine and was treated with rTMS of two frequencies (1 Hz and 10 Hz). The changes in cognitive function, brain structure, neurotrophic factor levels, and activity of miR-567/NEUROD2/PSD95 axis were assessed. The interaction between rTMS and miR-567 was further verified by inducing the level of miR-567 in AD mice.
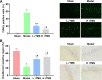
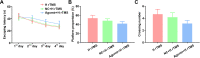
Results: The administrations of rTMS improved the cognitive function of AD mice and attenuated brain tissue destruction, which were associated with the restored production of BDNF and NGF. Additionally, rTMS administrations suppressed the expression of miR-567 and up-regulated the expressions of NEUROD2 and PSD95, which contributed to the improved condition in central nerve system. With the induced level of miR-567, the effects of rTMS were counteracted: the learning and memorizing abilities of mice were impaired, the brain neuron viability was suppressed, and the production of neurotrophic factors was suppressed even under the administration of rTMS. The changes in brain function and tissues were associated with the inhibited expressions of NEUROD2 and PSD95.
Conclusion: The findings outlined in the current study demonstrated that rTMS treatment could protect brain against AD-induced MCI without significant side effects, and the function depended on the inhibition of miR-567.
Keywords: Alzheimer’s disease; NEUROD2; miR-567; mild cognitive impairment; repetitive transcranial magnetic stimulation.
© 2021 Pang and Shi.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures
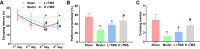

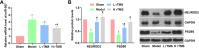

Similar articles
-
Protective effects of repetitive transcranial magnetic stimulation against 6-OHDA-induced Parkinson's symptoms in a mice model: the key role of miR-409-3p/PDHB axis.Int J Neurosci. 2023 May;133(5):578-585. doi: 10.1080/00207454.2021.1938037. Epub 2021 Jun 28. Int J Neurosci. 2023. PMID: 34078216
-
Administration of rTMS Alleviates Stroke-Induced Cognitive Deficits by Modulating miR-409-3p/CTRP3/AMPK/Sirt1 Axis.J Mol Neurosci. 2022 Mar;72(3):507-515. doi: 10.1007/s12031-021-01924-5. Epub 2021 Oct 16. J Mol Neurosci. 2022. PMID: 34655390
-
Administration of Repetitive Transcranial Magnetic Stimulation Attenuates Aβ 1-42-Induced Alzheimer's Disease in Mice by Activating β-Catenin Signaling.Biomed Res Int. 2019 Mar 5;2019:1431760. doi: 10.1155/2019/1431760. eCollection 2019. Biomed Res Int. 2019. PMID: 30949496 Free PMC article.
-
Cognitive Enhancement of Repetitive Transcranial Magnetic Stimulation in Patients With Mild Cognitive Impairment and Early Alzheimer's Disease: A Systematic Review and Meta-Analysis.Front Cell Dev Biol. 2021 Sep 10;9:734046. doi: 10.3389/fcell.2021.734046. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34568342 Free PMC article. Review.
-
Effects of Repetitive Transcranial Magnetic Stimulation in Patients With Mild Cognitive Impairment: A Meta-Analysis of Randomized Controlled Trials.Front Hum Neurosci. 2021 Oct 26;15:723715. doi: 10.3389/fnhum.2021.723715. eCollection 2021. Front Hum Neurosci. 2021. PMID: 34764859 Free PMC article.
Cited by
-
Therapeutic non-invasive brain treatments in Alzheimer's disease: recent advances and challenges.Inflamm Regen. 2022 Oct 3;42(1):31. doi: 10.1186/s41232-022-00216-8. Inflamm Regen. 2022. PMID: 36184623 Free PMC article. Review.
-
Epigenome Defines Aberrant Brain Laterality in Major Mental Illnesses.Brain Sci. 2024 Mar 7;14(3):261. doi: 10.3390/brainsci14030261. Brain Sci. 2024. PMID: 38539649 Free PMC article. Review.
-
Effects of transcranial magnetic stimulation on neurobiological changes in Alzheimer's disease (Review).Mol Med Rep. 2022 Apr;25(4):109. doi: 10.3892/mmr.2022.12625. Epub 2022 Feb 4. Mol Med Rep. 2022. PMID: 35119081 Free PMC article. Review.
-
Effect of transcranial magnetic stimulation in combination with citalopram on patients with post-stroke depression.Front Hum Neurosci. 2022 Oct 7;16:962231. doi: 10.3389/fnhum.2022.962231. eCollection 2022. Front Hum Neurosci. 2022. PMID: 36277050 Free PMC article.
-
Hippocampal differential expression underlying the neuroprotective effect of delta-9-tetrahydrocannabinol microdose on old mice.Front Neurosci. 2023 Jul 18;17:1182932. doi: 10.3389/fnins.2023.1182932. eCollection 2023. Front Neurosci. 2023. PMID: 37534036 Free PMC article.
References
-
- Manenti R, Cotelli M, Calabria M, Maioli C, Miniussi C. The role of the dorsolateral prefrontal cortex in retrieval from long-term memory depends on strategies: a repetitive transcranial magnetic stimulation study. Neuroscience. 2010;166(2):501–507. doi:10.1016/j.neuroscience.2009.12.037 - DOI - PubMed
-
- Manenti R, Tettamanti M, Cotelli M, Miniussi C, Cappa SF. The neural bases of word encoding and retrieval: a fMRI-guided transcranial magnetic stimulation study. Brain Topogr. 2010;22(4):318–332. - PubMed
LinkOut - more resources
Full Text Sources

