Regional specific differentiation of integumentary organs: SATB2 is involved in α- and β-keratin gene cluster switching in the chicken
- PMID: 34240503
- PMCID: PMC8742846
- DOI: 10.1002/dvdy.396
Regional specific differentiation of integumentary organs: SATB2 is involved in α- and β-keratin gene cluster switching in the chicken
Abstract
Background: Animals develop skin regional specificities to best adapt to their environments. Birds are excellent models in which to study the epigenetic mechanisms that facilitate these adaptions. Patients suffering from SATB2 mutations exhibit multiple defects including ectodermal dysplasia-like changes. The preferential expression of SATB2, a chromatin regulator, in feather-forming compared to scale-forming regions, suggests it functions in regional specification of chicken skin appendages by acting on either differentiation or morphogenesis.
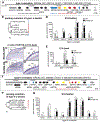
Results: Retrovirus mediated SATB2 misexpression in developing feathers, beaks, and claws causes epidermal differentiation abnormalities (e.g. knobs, plaques) with few organ morphology alterations. Chicken β-keratins are encoded in 5 sub-clusters (Claw, Feather, Feather-like, Scale, and Keratinocyte) on Chromosome 25 and a large Feather keratin cluster on Chromosome 27. Type I and II α-keratin clusters are located on Chromosomes 27 and 33, respectively. Transcriptome analyses showed these keratins (1) are often tuned up or down collectively as a sub-cluster, and (2) these changes occur in a temporo-spatial specific manner.
Conclusions: These results suggest an organizing role of SATB2 in cluster-level gene co-regulation during skin regional specification.
Keywords: epidermal differentiation; epigenetic mechanism; gene cluster; morphogenesis; regional specificity; α- and β-keratin.
© 2021 American Association for Anatomy.
Conflict of interest statement
Figures
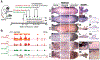






Similar articles
-
Regional Specific Differentiation of Integumentary Organs: Regulation of Gene Clusters within the Avian Epidermal Differentiation Complex and Impacts of SATB2 Overexpression.Genes (Basel). 2021 Aug 23;12(8):1291. doi: 10.3390/genes12081291. Genes (Basel). 2021. PMID: 34440465 Free PMC article.
-
Topographical mapping of α- and β-keratins on developing chicken skin integuments: Functional interaction and evolutionary perspectives.Proc Natl Acad Sci U S A. 2015 Dec 8;112(49):E6770-9. doi: 10.1073/pnas.1520566112. Epub 2015 Nov 23. Proc Natl Acad Sci U S A. 2015. PMID: 26598683 Free PMC article.
-
Genomic organization, transcriptomic analysis, and functional characterization of avian α- and β-keratins in diverse feather forms.Genome Biol Evol. 2014 Aug 24;6(9):2258-73. doi: 10.1093/gbe/evu181. Genome Biol Evol. 2014. PMID: 25152353 Free PMC article.
-
Avian skin development and the evolutionary origin of feathers.J Exp Zool B Mol Dev Evol. 2003 Aug 15;298(1):57-72. doi: 10.1002/jez.b.26. J Exp Zool B Mol Dev Evol. 2003. PMID: 12949769 Review.
-
Cytochemical and molecular characteristics of the process of cornification during feather morphogenesis.Prog Histochem Cytochem. 2008;43(1):1-69. doi: 10.1016/j.proghi.2008.01.001. Epub 2008 Mar 14. Prog Histochem Cytochem. 2008. PMID: 18394491 Review.
Cited by
-
Regional Specific Differentiation of Integumentary Organs: Regulation of Gene Clusters within the Avian Epidermal Differentiation Complex and Impacts of SATB2 Overexpression.Genes (Basel). 2021 Aug 23;12(8):1291. doi: 10.3390/genes12081291. Genes (Basel). 2021. PMID: 34440465 Free PMC article.
-
Analysis and comparison of protein secondary structures in the rachis of avian flight feathers.PeerJ. 2022 Feb 28;10:e12919. doi: 10.7717/peerj.12919. eCollection 2022. PeerJ. 2022. PMID: 35251779 Free PMC article.
References
-
- Chuong CM. Molecular Basis of Epithelial Appendage Morphogenesis. Austin, TX: Landes Bioscience; 1998.
-
- Jiang T, Stott N, Widelitz R, Chuong CM. Current methods in the Study of Avian Skin Appendages. In: Chuong CM, ed. Molecular basis of epithelial appendage morphogenesis. Austin, TX: Landes Bioscience; 1998:395–408.
-
- Millar SE. Molecular mechanisms regulating hair follicle development. J Invest Dermatol. 2002;118(2):216–25. https://doi.org10.1046/j.0022-202x.2001.01670.x. - PubMed
-
- Wu P, Hou LH, Plikus M, et al. Evo-Devo of amniote integuments and appendages. International Journal of Developmental Biology. 2004;48(2-3):249–270. https://doi.orgDOI 10.1387/ijdb.15272390. - DOI - PMC - PubMed
-
- Chang WL, Wu H, Chiu YK, et al. The Making of a Flight Feather: Bio-architectural Principles and Adaptation. Cell. 2019;179(6):1409–1423 e17. https://doi.org10.1016/j.cell.2019.11.008. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

