Differential polarization and activation dynamics of systemic T helper cell subsets after aneurysmal subarachnoid hemorrhage (SAH) and during post-SAH complications
- PMID: 34244562
- PMCID: PMC8270974
- DOI: 10.1038/s41598-021-92873-x
Differential polarization and activation dynamics of systemic T helper cell subsets after aneurysmal subarachnoid hemorrhage (SAH) and during post-SAH complications
Abstract
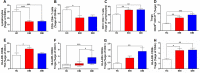
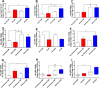
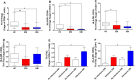
Aneurysmal subarachnoid hemorrhage (SAH) is associated with high morbidity and mortality. Devastating post-SAH complications, such as cerebral vasospasm (CVS), delayed cerebral ischemia or seizures to mention a few, are mainly responsible for the poor clinical outcome. Inflammation plays an indispensable role during early brain injury (EBI) and delayed brain injury (DBI) phases over which these complications arise. T helper cells are the major cytokine secreting cells of adaptive immunity that can polarize to multiple functionally unique sub-populations. Here, we investigate different CD4+ T cell subsets during EBI and DBI phases after SAH, and their dynamics during post-SAH complications. Peripheral venous blood from 15 SAH patients during EBI and DBI phases, was analyzed by multicolour flowcytometry. Different subsets of CD3+ CD4+ T cells were characterized by differential cell surface expression of CXCR3 and CCR6 into Th1, Th2, Th17, whereas Tregs were defined by CD25hiCD127lo. The analysis of activation states was done by the expression of stable activation markers CD38 and HLA-DR. Interestingly, compared to healthy controls, Tregs were significantly increased during both EBI and DBI phases. Different activation states of Tregs showed differential significant increase during EBI and DBI phases compared to controls. HLA-DR- CD38+ Tregs were significantly increased during DBI phase compared to EBI phase in SAH patients developing CVS, seizures and infections. However, HLA-DR- CD38- Tregs were significantly reduced during EBI phase in patients with cerebral ischemia (CI) compared to those without CI. HLA-DR- CD38- Th2 cells were significantly increased during EBI phase compared to controls. A significant reduction in Th17/Tregs and HLA-DR- CD38+ Th17/Tregs ratios was observed during both EBI and DBI phases compared to controls. While HLA-DR- CD38- Th17/Tregs and HLA-DR- CD38- Th1/Th2 ratios were impaired only during EBI phase compared to controls. In conclusion, CD4+ T cell subsets display dynamic and unique activation patterns after SAH and during the course of the manifestation of post-SAH complications, which may be helpful for the development of precision neurovascular care. However, to claim this, confirmatory studies with larger patient cohorts, ideally from different ethnic backgrounds, are required. Moreover, our descriptive study may be the grounds for subsequent lab endeavors to explore the underlying mechanisms of our observations.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
NLRP3 inhibition attenuates early brain injury and delayed cerebral vasospasm after subarachnoid hemorrhage.J Neuroinflammation. 2021 Jul 20;18(1):163. doi: 10.1186/s12974-021-02207-x. J Neuroinflammation. 2021. PMID: 34284798 Free PMC article.
-
The Role of Platelet Activation and Inflammation in Early Brain Injury Following Subarachnoid Hemorrhage.Neurocrit Care. 2017 Feb;26(1):48-57. doi: 10.1007/s12028-016-0292-4. Neurocrit Care. 2017. PMID: 27430874 Free PMC article.
-
Toll-like receptor-4 pathway as a possible molecular mechanism for brain injuries after subarachnoid hemorrhage.Int J Neurosci. 2020 Sep;130(9):953-964. doi: 10.1080/00207454.2019.1709845. Epub 2020 Jan 14. Int J Neurosci. 2020. PMID: 31903827 Review.
-
HLA-DR expression on regulatory T cells is closely associated with the global immune activation in HIV-1 infected subjects naïve to antiretroviral therapy.Chin Med J (Engl). 2011 Aug;124(15):2340-6. Chin Med J (Engl). 2011. PMID: 21933566
-
Advances in experimental subarachnoid hemorrhage.Acta Neurochir Suppl. 2011;110(Pt 1):15-21. doi: 10.1007/978-3-7091-0353-1_3. Acta Neurochir Suppl. 2011. PMID: 21116908 Review.
Cited by
-
Remote Ischemic Post-conditioning Reduces Cognitive Impairment in Rats Following Subarachnoid Hemorrhage: Possible Involvement in STAT3/STAT5 Phosphorylation and Th17/Treg Cell Homeostasis.Transl Stroke Res. 2025 Jun;16(3):600-611. doi: 10.1007/s12975-024-01235-y. Epub 2024 Feb 15. Transl Stroke Res. 2025. PMID: 38356020
-
Regulatory T Cell- and Natural Killer Cell-Mediated Inflammation, Cerebral Vasospasm, and Delayed Cerebral Ischemia in Aneurysmal Subarachnoid Hemorrhage-A Systematic Review and Meta-Analysis Approach.Int J Mol Sci. 2025 Feb 1;26(3):1276. doi: 10.3390/ijms26031276. Int J Mol Sci. 2025. PMID: 39941044 Free PMC article.
-
Regulatory Role of Mesenchymal Stem Cells on Secondary Inflammation in Spinal Cord Injury.J Inflamm Res. 2022 Jan 26;15:573-593. doi: 10.2147/JIR.S349572. eCollection 2022. J Inflamm Res. 2022. PMID: 35115806 Free PMC article. Review.
-
Prognostic value of platelet levels in patients with aneurysmal Subarachnoid Hemorrhage.Sci Rep. 2024 Jul 20;14(1):16743. doi: 10.1038/s41598-024-67322-0. Sci Rep. 2024. PMID: 39033250 Free PMC article.
-
Immunological Profile of Vasospasm after Subarachnoid Hemorrhage.Int J Mol Sci. 2023 May 16;24(10):8856. doi: 10.3390/ijms24108856. Int J Mol Sci. 2023. PMID: 37240207 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

